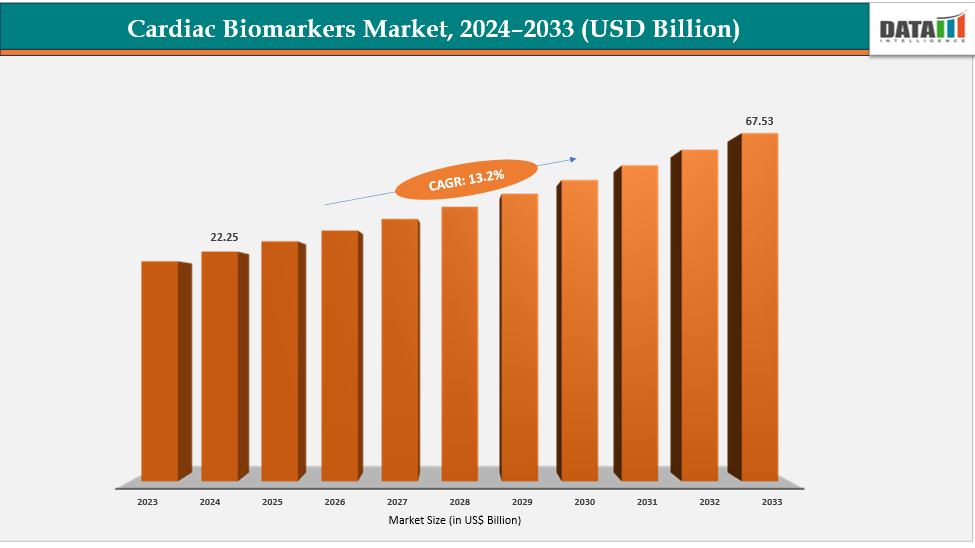
Global Cardiac Biomarkers Market Size
Global Cardiac Biomarkers Market reached US$ 25.20 billion in 2025 and is expected to reach US$ 73.28 billion by 2033, growing at a CAGR of 13.22% during the forecast period 2026-2033.
The cardiac biomarkers market is being driven mostly by rising awareness of early diagnosis and preventative healthcare. Tests including CK-MB, BNP, and high-sensitivity troponin are in greater demand as patients and healthcare professionals place a higher priority on routine screening for cardiovascular risk factors. Patient outcomes are improved when myocardial infarction, heart failure, and acute coronary syndrome are detected early enough to allow for prompt intervention. Market expansion is further supported by technology developments in multiplex and point-of-care assays, as well as public and corporate programs encouraging preventative care. Modern healthcare is becoming increasingly dependent on cardiac biomarker testing due to the increasing incidence of cardiovascular disorders and proactive patient behaviour.
Market Dynamics
Drivers:Rising prevalence of cardiovascular diseases are significantly driving the cardiac biomarkers market growth
The rising prevalence of cardiovascular diseases (CVDs) is a key driver of the cardiac biomarkers market. Rapid and precise diagnostics are now more important than ever because of the rise in heart attacks, heart failure, and acute coronary syndromes brought on by aging populations, diabetes, obesity, and sedentary lifestyles. Troponins, CK-MB, and BNP are examples of cardiac biomarkers that are crucial for early diagnosis, risk assessment, and monitoring in both normal and emergency situations. Global adoption of high-sensitivity assays and point-of-care cardiac biomarker kits has increased dramatically as a result of governments, hospitals, and insurers prioritizing improved diagnostic tools due to rising healthcare expenditures and mortality from CVDs.
Restraints: The high cost of advanced assays and devices of cardiac biomarkers tests are hampering the growth of the cardiac biomarker market.
The high cost of advanced cardiac biomarker assays and devices is a major factor restraining market growth. High-sensitivity troponin tests, BNP assays, and point-of-care devices require significant investment, making them less accessible in low- and middle-income countries. Low insurance reimbursement further raises patients' costs and discourages them from getting regular screenings or follow-up exams. The widespread adoption of expensive equipment is slowed by smaller hospitals and rural clinics' frequent avoidance of it. Costlier substitutes like imaging and ECG are also favored in environments with limited resources. This disparity in pricing limits the number of tests that can be performed and postpones widespread acceptance, which eventually hinders the market expansion for cardiac biomarkers.
Competitive Landscape
Top companies in the Cardiac Biomarkers Market are Roche Diagnostics Limited, Abbott, Siemens Healthineers AG, Thermo Fisher Scientific Inc., AdvaCare Pharma, Novus Biologicals, Fine Biotech Co., Ltd., Labome, Vitrosens Biyoteknoloji, and Real-Gene Labs, among others.
Roche Diagnostics Limited: Roche Diagnostics is a global leader in cardiac biomarker testing, offering high-sensitivity assays such as Elecsys Troponin T Gen 5 and Elecsys proBNP II, along with the cobas h 232 point-of-care system. These solutions enable rapid, accurate diagnosis and management of myocardial infarction and heart failure across laboratories, hospitals, and point-of-care settings.
Key Developments
March 2026: Roche Diagnostics launched an advanced high-sensitivity cardiac troponin assay with AI-enhanced algorithms for faster and more precise detection of acute myocardial infarction, improving point-of-care diagnostics worldwide.
February 2026: Abbott introduced a next-generation NT-proBNP test kit optimized for heart failure monitoring, featuring multiplex biomarker analysis for better prognostic accuracy.
January 2026: Siemens Healthineers unveiled a novel CK-MB and myoglobin combo test with improved specificity for early myocardial injury detection, leveraging nanotechnology for superior sensitivity.
Market Scope
| Metrics | Details | |
| CAGR | 13.22% | |
| Market Size Available for Years | 2023-2033 | |
| Estimation Forecast Period | 2026-2033 | |
| Revenue Units | Value (US$ Bn) | |
| Segments Covered | By Cardiac Biomarker Tests | Troponin, Creatine kinase, Myoglobin, Galectin-3 and Other |
| By Application | Myocardial Infarction, Congestive Heart Failure, Acute Coronary Syndrome, Angina and Other | |
| By Testing Location | Laboratory Testing, Point of Care Testing | |
| Regions Covered | North America, Europe, Asia-Pacific, South America and the Middle East & Africa | |
The global cardiac biomarkers market report delivers a detailed analysis with 62 key tables, more than 55 visually impactful figures, and 159 pages of expert insights, providing a complete view of the market landscape.
For more pharmaceutical-related reports, please click here