Gene Therapy Market Size
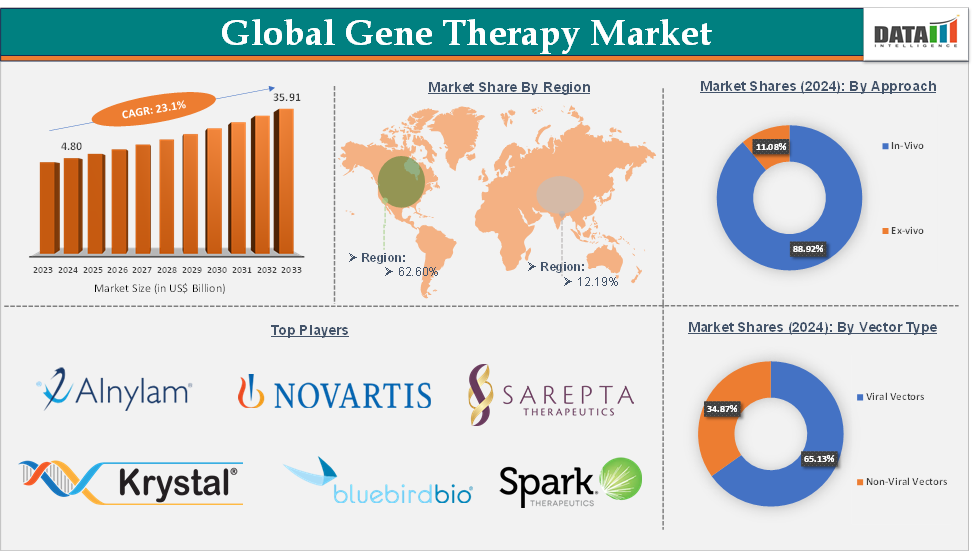
The Gene Therapy Market reached US$ 5.91 billion in 2025 and is expected to reach US$ 44.20 billion by 2033, growing at a CAGR of 23.1% during the forecast period 2026-2033.
Gene Therapy Market Overview
The gene therapy market is experiencing rapid growth, driven by advancements in genomics, biotechnology and personalized medicine. Gene therapy is revolutionizing genetic disorders, rare diseases and cancer treatments. The market has witnessed significant expansion due to the increasing R&D investments, strategic partnerships among key players and approval of novel gene therapies in emerging markets.
Executive Summary
For more details on this report – Request for Sample
Gene Therapy Market Dynamics: Drivers & Restraints
Expansion into the rare diseases is significantly driving the gene therapy market growth
Rare diseases, which are genetic in nature, often have limited or no effective treatment options, making them ideal targets for gene therapies. The unique nature of these diseases presents a substantial opportunity for gene therapy to provide curative treatments where traditional approaches fail. For instance, Luxturna, a gene therapy treating children and adult patients with an inherited form of vision loss that may result in blindness. Luxturna is the first directly administered gene therapy approved in the U.S. that targets a disease caused by mutations in a specific gene.
Spinal Muscular Atrophy (SMA) is a rare genetic disorder that leads to progressive muscle weakness. Zolgensma, a gene therapy, offers a one-time treatment that has demonstrated significant improvements in survival and motor function for children with SMA. Hemophilia, another rare genetic disorder, requires lifelong treatment with clotting factor concentrates.
For instance, in February 2025, CSL Behring announced the four-year results from the pivotal HOPE-B study confirming the long-term durability and safety of a one-time infusion of HEMGENIX (etranacogene dezaparvovec-drlb) gene therapy for adults living with hemophilia B. As more gene therapies for rare diseases gain approval, the market will see a diversification of therapies, addressing a wider range of conditions.
High cost associated with the gene therapies are hampering the market growth
The high cost of gene therapies is one of the most significant challenges impacting the growth and accessibility of the gene therapy market. While gene therapies have shown curative potential for many serious diseases, their expensive price tags pose barriers to widespread adoption. For instance, the gene therapies Zynteglo and Skysona are priced at US$ 2.8 million and US$ 3 million per dose. Moreover, Zolgensma has a reported list price of US$ 2.1 million.
The development of gene therapies is an expensive, lengthy process. Clinical trials for gene therapies often span several years and involve high patient monitoring and expensive regulatory approval procedures. For instance, Luxturna, a gene therapy for Leber’s congenital amaurosis, took over 10 years from development to approval, and its cost is $850,000 per patient.
Many gene therapies are personalized treatments that are tailored to the genetic makeup of individual patients, requiring bespoke production for each case. This individualization further increases costs. For instance, CAR-T therapies such as Yescarta and Kymriah are customized for each patient by collecting and modifying the patient’s own T-cells, a process that involves complex manufacturing and quality control measures. These therapies can cost over $300,000 to $400,000 per patient.
Gene Therapy Market, Segment Analysis
The global gene therapy market is segmented based on approach, vector type, technique, application, and region.
Vector Type:
The viral vectors segment is expected to hold 65.13% of the market share in 2024 in the gene therapy market
Viral vectors are modified viruses used as delivery vehicles to introduce genetic material into cells for gene therapy. They exploit viruses' natural ability to transport their genomes into host cells, a process called transduction. The choice of viral vector depends on factors such as efficiency, safety, toxicity, stability, and the desired duration of transgene expression.
Currently, there are four types of viral vectors used in gene therapy: adenovirus, adeno-associated virus (AAV), lentivirus, and gamma-retrovirus (γ-retroviral). Each vector type requires a complex manufacturing process. These vectors are chosen for their ability to efficiently deliver genetic material, each having its strengths and weaknesses depending on the specific therapeutic application.
Gene Therapy Market Top Companies
Top companies in the gene therapy market include Alnylam Pharmaceuticals, Inc., Spark Therapeutics, Inc., Novartis AG, bluebird bio, Inc., Ferring Pharmaceuticals Inc., Vertex Pharmaceuticals Incorporated, Sarepta Therapeutics, Inc., CSL Behring LLC, Amgen, Inc., Orchard Therapeutics group., Krystal Biotech, Inc., and among others.
Recent Developments
- April 2026 – Novartis, Vertex Pharmaceuticals, and Sarepta Therapeutics expand late-stage gene therapy pipelines
Leading biotech firms are advancing Phase 2/3 gene therapies targeting rare genetic disorders, neuromuscular diseases, and hematological conditions, accelerating commercialization timelines. - March 2026 – bluebird bio and CSL Behring focus on hematology and rare disease gene therapies
Companies are prioritizing treatments for beta-thalassemia, sickle cell disease, and hemophilia, with ongoing clinical trials and commercialization efforts. - March 2026 – Spark Therapeutics and Krystal Biotech advance AAV-based gene therapies
Adeno-associated virus (AAV) vectors remain a leading platform, supporting treatments for ophthalmology, dermatology, and rare genetic diseases. - February 2026 – Amgen and Ferring Pharmaceuticals expand investments in gene therapy R&D
Large pharma companies are increasing funding and partnerships to strengthen internal gene therapy pipelines and manufacturing capabilities. - February 2026 – Growth in in vivo gene editing and CRISPR-based therapies
Gene editing technologies, including CRISPR, are advancing rapidly, enabling precise, one-time treatments for previously untreatable genetic disorders. - January 2026 – Orchard Therapeutics and Alnylam Pharmaceuticals expand rare disease treatment portfolios
Companies are focusing on ultra-rare genetic disorders, leveraging gene therapy and RNA-based approaches to address unmet medical needs.
Market Scope
| Metrics | Details | |
| CAGR | 23.1% | |
| Market Size Available for Years | 2023-2033 | |
| Estimation Forecast Period | 2026-2033 | |
| Revenue Units | Value (US$ Bn) | |
| Segments Covered | Approach | Ex-Vivo and In-Vivo |
| Vector Type | Viral Vectors and Non-Viral Vectors | |
| Technique | Gene Addition, Gene Silencing, and Gene Editing | |
| Application | Oncology, Musculoskeletal Conditions, Blood Disorders, Rare Diseases, Ophthalmology, and Others | |
| Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
The global gene therapy market report delivers a detailed analysis with 73 key tables, more than 64 visually impactful figures, and 168 pages of expert insights, providing a complete view of the market landscape.