Wound Care Biologics Market Size
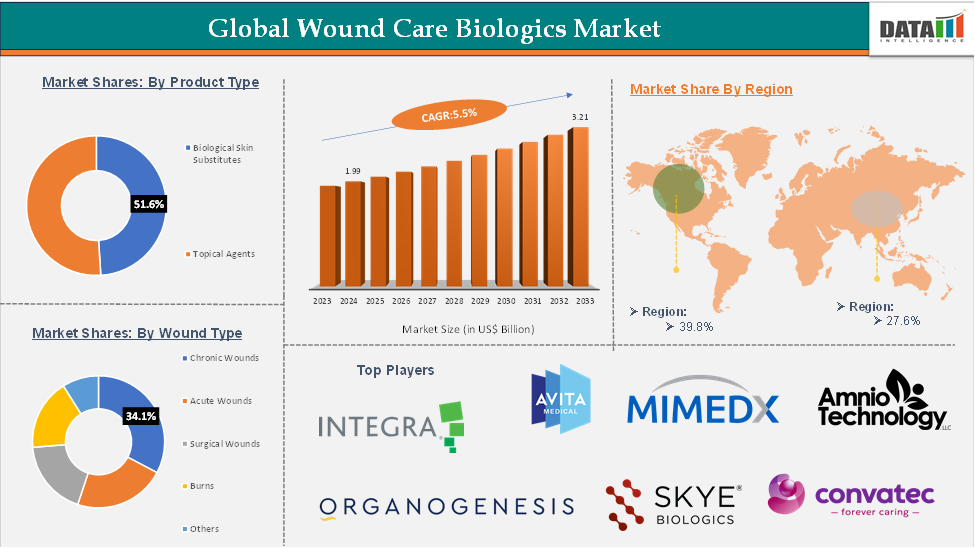
Wound Care Biologics Market size reached US$ 1.99 Billion in 2024 and is expected to reach US$ 3.21 Billion by 2033, growing at a CAGR of 5.5% during the forecast period 2025-2033.
The global wound care biologics market is witnessing robust growth, driven by the rising prevalence of chronic wounds, the increasing geriatric population, and advancements in biologic therapies. Biologic wound care products are gaining traction for their ability to accelerate healing, reduce infection risks, and improve clinical outcomes in patients suffering from complex wounds, including diabetic ulcers, pressure sores, and burn injuries.
North America continues to lead the market, owing to its strong healthcare infrastructure, high disease burden, and active pipeline of innovative product launches. Companies across the region are focusing on R&D, strategic collaborations, and international expansion to address this growing demand.
Executive Summary
For more details on this report - Request for Sample
Wound Care Biologics Market Dynamics: Drivers & Restraints
Rising prevalence of chronic diseases is expected to drive the wound care biologics market
The rising incidence of diseases, such as diabetes and burn injuries, is expected to boost the market over the forecast period. According to an article published in Star Pearls in May 2022, the annual incidence of diabetic foot ulcers worldwide is between 9.1 and 26.1 million. Around 15 to 25% of patients with diabetes mellitus will develop a diabetic foot ulcer in their lifetime.
As the number of newly diagnosed diabetics is increasing annually, the incidence of diabetic foot ulcers is also bound to rise. Diabetic foot ulcers can arise at any age, despite being most prevalent in patients aged 45 and over with diabetes mellitus.
Patients suffering from diabetes mellitus develop intense atherosclerosis of the small blood vessels in the legs and feet, leading to vascular compromise, which is another cause of diabetic foot infections. Healing is delayed, eventually leading to necrosis and gangrene because blood cannot reach the wound. The rising disease incidence leads to an increasing demand for wound care biologics worldwide.
High costs of wound care biologics are expected to hinder the market
The high costs associated with wound care biologics are expected to act as a significant barrier to the growth of the market. Biologic products, such as growth factors, amniotic membranes, and cell-based therapies, offer advanced treatment options for chronic and complex wounds, but they come at a premium price compared to traditional wound care methods..
Wound Care Biologics Market Segment Analysis
The global wound care biologics market is segmented based on product type, wound type, end user, and region.
Wound Type:
The chronic wounds segment is expected to hold 51.6% of the global wound care biologics market
Chronic wounds pose a persistent and serious health challenge, especially for individuals with underlying conditions such as diabetes, obesity, and vascular disorders, which significantly delay the healing process. These wounds are not only painful and slow to heal but also increase the risk of infections, sepsis, and even amputations if left untreated.
According to the American Academy of Home Care Medicine, in the United States alone, an estimated 6.5 million people are affected by chronic wounds. This highlights the widespread nature of the condition and the burden it places on healthcare systems.
The chronic wounds segment is currently the dominant segment in the wound care biologics market, largely due to the high prevalence of such conditions and the growing need for advanced, effective treatment options. Biologic products such as growth factors, skin substitutes, and bioengineered dressings are increasingly being used in the treatment of chronic wounds because of their ability to accelerate healing, reduce infection risks, and improve tissue regeneration.
Additionally, companies are forming strategic alliances to innovate and provide advanced biologic solutions for chronic wounds. For instance, in February 2025, Royal Wound-X, a division of Royal Biologics dedicated to advancing wound care solutions, announced a strategic partnership with Coldplasmatech GmbH, a leader in medical cold plasma technology. Together, they aim to revolutionize the treatment of complex and chronic wounds globally.
Thus, the aging population, rising incidence of lifestyle-related chronic diseases, and ongoing advancements in wound care biologics specifically target the complex nature of chronic wound healing, are expected to hold the segment in the dominant position.
Wound Care Biologics Market Geographical Analysis
North America is expected to hold 39.8% of the global wound care biologics market
North America maintains its leadership in the wound care biologics market, driven by a combination of demographic trends, technological advancements, and strategic industry initiatives. The region's robust healthcare infrastructure, coupled with a high prevalence of chronic conditions such as diabetes and vascular diseases, has led to an increased demand for advanced wound care solutions, positioning biologics as a critical component for treatment protocols.
In addition to demographic factors, strategic initiatives by key market players are reinforcing North America's strong position in the market. For instance, in April 2025, LifeNet Health announced the launch of Dermacell Porous, an advanced human acellular dermal matrix designed to retain growth factors, collagen, and elastin in a biohospitable scaffold. This innovation aims to improve wound healing, particularly for underserved communities affected by diabetes, where access to quality healthcare is limited. LifeNet Health continues its mission to save lives and restore health through regenerative medicine.
Furthermore, in October 2024, Royal Biologics introduced two innovative products at a symposium on advanced wound care: Peak Powder Collagen Matrix, which enhances healing by creating a gel-like barrier that supports tissue regeneration, and ElectroFiber 3D, a bioengineered matrix that promotes cellular migration and reduces healing times.
These developments underscore North America's proactive approach in advancing wound care biologics and meeting the growing clinical demand. The region's commitment to innovation, coupled with a supportive regulatory environment, positions it well to continue leading the global wound care biologics market.
Wound Care Biologics Market Top Companies
The top companies in the wound care biologics market include Organogenesis Inc., Integra Lifesciences, Convatec Inc., Mimedx, Skye Biologics, Amnio Technology, LLC, Dynamic Medical Services DBA Acesso Biologics, PolyNovo Limited, VIVEX Biologics, Inc., Zimmer Biomet, among others.
Key Developments
In January 2025, BioStem Technologies announced the initiation of the BR-AM-DFU (BioREtain Amniotic Membrane - Diabetic Foot Ulcers) clinical trial, aimed at evaluating the effectiveness of Vendaje in the treatment of non-healing diabetic foot ulcers. This study represents a significant step in advancing biologic therapies for chronic wound management, particularly for patients with diabetes-related complications.
Market Scope
Metrics | Details | |
CAGR | 5.5% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Bn) | |
Segments Covered | Product Type | Biological Skin Substitutes, Topical Agents |
Wound Type | Chronic Wounds, Acute Wounds, Surgical Wounds, Burns, Others | |
End User | Hospitals, Ambulatory Surgical Centers, Others | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
The global wound care biologics market report delivers a detailed analysis with 57 key tables, more than 46 visually impactful figures, and 168 pages of expert insights, providing a complete view of the market landscape.