Next Generation Sequencing (NGS) Market Size
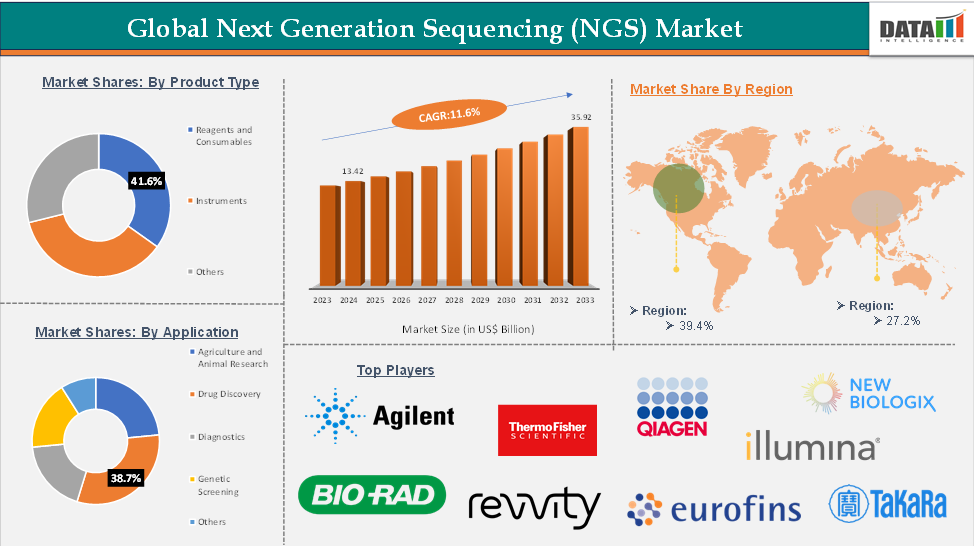
Next Generation Sequencing (NGS) market size reached US$15.2 billion in 2025 and is expected to reach US$35.92 billion by 2033, growing at a CAGR of 11.6% during the forecast period 2026-2033.
Sequencing on NGS includes several significant processes. For instance, DNA next-generation sequencing (NGS) calls for library preparation, bioinformatics analysis, DNA fragmentation, enormous parallel sequencing, and variant/mutation identification and interpretation. Furthermore, rising consumer demand for NGS due to the rising availability of products and technologies, an increase in the number of authorized service providers in the market, and an increase in drug discovery research are driving up the next-generation sequencing (NGS) market size.
The market is experiencing a growing demand for products from North American areas due to the rising advancements in this field. Significant competitors like Thermo Fisher Scientific Inc., Illumina, Inc., Bio-Rad Laboratories, Inc., and others are actively operating in the market.
Executive Summary
For more details on this report – Request for Sample
Next Generation Sequencing (NGS) Market Dynamics: Drivers & Restraints
The advancements in NGS platforms are expected to drive the market
The increase in demand for next-generation sequencing (NGS) for various applications, novel technology launches, recent developments in therapeutics, surging research, and novel product launches are likely to fuel the global next-generation sequencing (NGS) market size expansion throughout the forecast period.
For instance, in February 2025, Roche launched its proprietary Sequencing by Expansion (SBX) technology, a breakthrough in next-generation sequencing that delivers ultra-fast, high-throughput, and scalable genomic analysis. This innovation enhances the ability to decode complex diseases like cancer and neurodegenerative disorders by providing deeper insights into gene functions and interactions.
Nucleic acid sequencing has benefited from technological advancements in terms of technical proficiency. For instance, in April 2024, NewBiologix SA launched its next-generation sequencing (NGS) and optical mapping platform. This advanced suite of integrated technologies is designed to provide comprehensive genomic analysis services, specifically tailored to meet the needs of the biopharmaceutical industry. Thus, advancements in NGS platforms are playing a pivotal role in driving the growth of the NGS market by enabling more comprehensive, accurate, and efficient genomic analysis for the biopharmaceutical industry.
Limitations associated with NGS technology are expected to hinder the next-generation sequencing (NGS) market
The idea of using NGS in ordinary clinical practice is difficult since these technologies can successfully identify clinically significant mutations, but they frequently fail to do so when larger sections of the genome are analyzed. The determination of the appropriate quality parameters, for example, could significantly aid in resolving these issues.
NGS technology will undoubtedly need measures for data management, process standardization and interpretation when it is introduced as a therapeutic tool. For the analysis of the enormous amount of data that these systems will produce, more emphasis should be placed on the work of biostatisticians and bioinformaticians.
Next Generation Sequencing (NGS) Market Segment Analysis
The global next-generation sequencing (NGS) market is segmented based on product type, application, technology, end user, and region.
The drug discovery segment is increasingly driving the growth of the next-generation sequencing (NGS) market, as evidenced by strategic initiatives from leading companies. For instance, in December 2024, Alithea Genomics SA launched its MERCURIUS Full-Length DRUG-seq library preparation technology, uniquely integrating large-scale sample multiplexing, an extraction-free workflow, and full transcript coverage. Designed to streamline high-throughput RNA sequencing, this technology enables up to 384 full-length RNA-seq library preparations in a single tube, making it particularly valuable for accelerating transcriptomic profiling in drug screening and target validation.
Similarly, in September 2024, Takara Bio Europe introduced the Shasta Single-Cell System at the 2024 Human Cell Atlas General Meeting, a high-throughput NGS platform designed to advance drug discovery by enabling novel biomarker identification in oncology research. This innovative system integrates robust chemistries with intuitive bioinformatics tools, allowing researchers to analyze genomic and transcriptomic data from significantly larger numbers of cells, thereby enhancing the efficiency and scalability of early-stage drug and biomarker discovery efforts. These developments are expected to increase the demand which are used in drug discovery, which further increases the segment’s proportion in the overall market growth, making it a dominant segment.
Next Generation Sequencing (NGS) Market Geographical Analysis
The growing demand for next-generation sequencing (NGS) in healthcare research is creating significant opportunities for manufacturers to expand operations across North America. With a large concentration of producers and suppliers, combined with rapid economic growth, the region has seen increased industrial output, which in turn is driving the uptake of NGS technologies. For instance, in October 2024, Illumina recently launched the MiSeq i100 Series, a new line of compact, user-friendly sequencing systems designed to make next-generation sequencing more accessible. The MiSeq i100 and i100 Plus offer rapid turnaround, affordable operation, and room-temperature reagent storage, helping labs start sequencing immediately
Furthermore, collaborative research initiatives between healthcare organizations and industry players, along with continuous innovation in product development aimed at enhancing diagnostic and treatment capabilities, are fueling further demand. Thus, North America continues to strengthen its dominant position in the global NGS market.
The Asia-Pacific region is expected to be the fastest-growing market in the next-generation sequencing (NGS) sector due to a combination of rising healthcare investments, large and genetically diverse populations, and strong government initiatives. The region’s vast population provides a rich resource for genetic research, driving demand for personalized medicine, diagnostics, and targeted therapies.
Next Generation Sequencing (NGS) Market Top Companies
The top companies in the next-generation sequencing (NGS) market include Thermo Fisher Scientific Inc., Agilent Technologies, Inc., Bio-Rad Laboratories, Inc., Qiagen, Eurofins Scientific, Revvity (PerkinElmer Inc.), Takara Bio Inc., Illumina Inc., Alithea Genomics SA, and NewBiologix SA, among others.
Recent Developments
February 2026: Across North America, Europe, and Asia Pacific, the increasing demand for precision medicine, rising prevalence of genetic disorders, and growing applications in oncology significantly boosted the next-generation sequencing market.
January 2026: Globally, advancements in high-throughput sequencing technologies and genomics enabled faster, more accurate, and cost-effective DNA and RNA analysis, expanding clinical and research applications.
December 2025: Leading biotechnology companies such as Illumina, Thermo Fisher Scientific, and BGI Genomics expanded their sequencing platforms and services through innovation and strategic collaborations.
November 2025: Increasing adoption of NGS in oncology, infectious disease diagnostics, reproductive health, and drug discovery improved disease detection, personalized treatment, and research outcomes.
October 2025: Companies focused on developing portable and benchtop sequencing systems, reducing turnaround time and enabling decentralized testing in clinical and research settings.
September 2025: Across key regions including the United States, Germany, China, India, and Japan, rising investments in genomic research, government funding, and expanding healthcare infrastructure accelerated market growth.
August 2025: In the United States, strong presence of advanced research institutions and increasing adoption of precision medicine significantly drove NGS market expansion.
July 2025: In Japan, growing focus on genomic research, aging population healthcare needs, and advancements in biotechnology supported the growth of the NGS market.
Market Scope
| Metrics | Details | |
| CAGR | 11.6% | |
| Market Size Available for Years | 2025-2033 | |
| Estimation Forecast Period | 2026-2033 | |
| Revenue Units | Value (US$ Bn) | |
| Segments Covered | Product Type | Reagents and Consumables, Instruments, Others |
| Application | Agriculture and Animal Research, Drug Discovery, Diagnostics, Genetic Screening, Others | |
| Technology | Whole Genome Sequencing, Chip Sequencing, De Novo Sequencing, RNA Sequencing, Methyl Sequencing, Others | |
| End User | Hospitals and Diagnostic Centers, Biotechnology and Pharmaceutical Companies, Academic & Research Institutes, Others | |
| Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
The global next-generation sequencing (NGS) market report delivers a detailed analysis with 57 key tables, more than 46 visually impactful figures, and 168 pages of expert insights, providing a complete view of the market landscape.