Disease Overview:
The Metabolic Dysfunction-Associated Steatohepatitis (MASH) Market size reached US$ 2.12 billion in 2025 and is expected to reach US$ 28.45 billion by 2033, growing at a CAGR of 38.2% during the forecast period 2026-2033.
Metabolic Dysfunction-Associated Steatohepatitis (MASH), formerly known as Nonalcoholic Steatohepatitis (NASH) is a disease caused by a build-up of fat in the liver, not caused by alcohol consumption. As a result of fat deposition, the liver becomes inflamed (hepatitis). The inflammation and liver damage from MASH can cause fibrosis and scarring and can lead to cirrhosis, where the liver is scarred and significantly damaged, often permanently. The development of MASH is linked with underlying conditions such as metabolic syndrome, obesity, and type 2 diabetes.
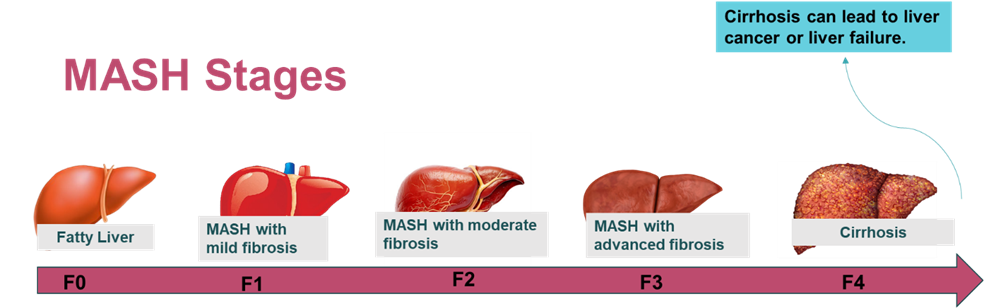
MASH is an advanced form of MSALD (Metabolic dysfunction-associated steatotic liver disease) and is staged based on the level of fibrosis observed in liver. Stage F0 refers to the early stage of MASH, where no scarring is observed, stage F1 refers to mild to moderate perisinusoidal or periportal fibrosis, stage F2 refers to both perisinusoidal and portal/periportal fibrosis, stage F3 refers to advanced fibrosis, and F4 refers to liver cirrhosis, which can progress to hepatocellular carcinoma and liver failure.
For more details on this report – Request for Sample
Diagnosis:
The diagnosis of MASH currently involves the following tests:
- Liver Stiffness Test: This is a non-invasive test for measuring liver elasticity and fibrosis, perfomed using the gold standard imaging techniques like transient elastography (FibroScan) or magnetic resonance elastography (MRE).
- Blood Tests: The common blood tests involve liver function tests for measuring liver enzymes such as ALT (alanine aminotransferase) and AST (aspartate aminotransferase). fibrosis assessment tests such as AST-to-Platelet Ratio Index (APRI) score and Fibrosis-4 (Fib-4) score for measuring the level of liver scarring or fibrosis, and lipid panel.
- Liver Biopsy: This is the gold standard confirmatory test for MASH, which involves microscopic observation of collected liver tissue for fibrosis and scarring. The downside of it is its invasive nature.
Treatment:
The current standard of care of MASH involves the combination of lifestyle interventions with bariatric surgery and pharmacotherapy to address the underlying disease. However, In 2024, Rezdiffra (resmetirom) became the first drug to be approved by the U.S. FDA. Below is the current treatment options available for MASH treatment.
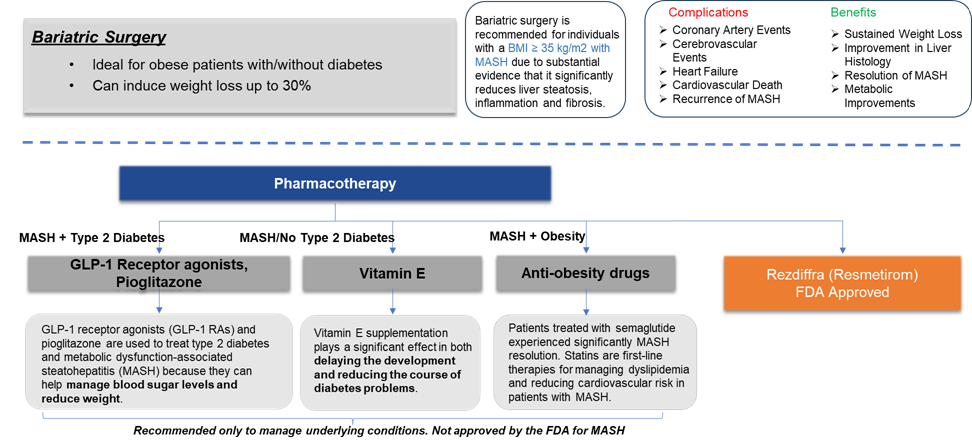
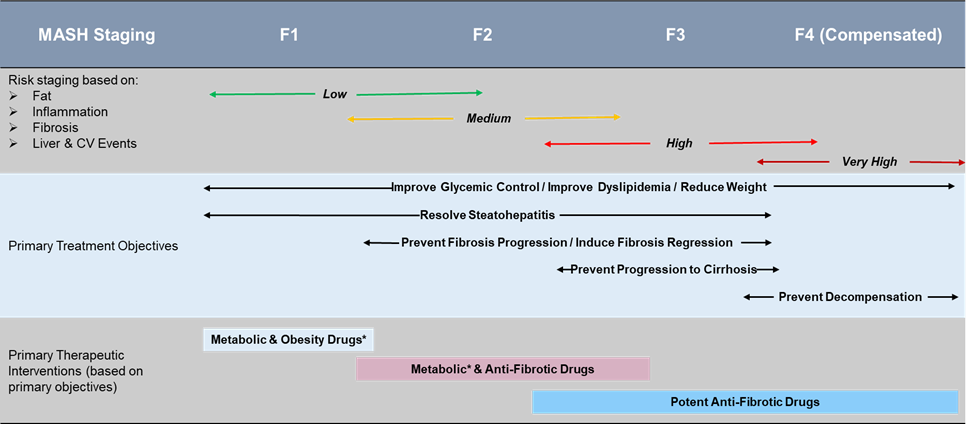
However, the treatment goals differ as per the stage of diagnosis. The treatment for stage F0 usually involves lifestyle modifications such as diet and exercise, along with addressing any underlying condition. Likewise, the treatment goals vary for each stage as mentioned below.
Metabolic Dysfunction-Associated Steatohepatitis Market Executive Summary:
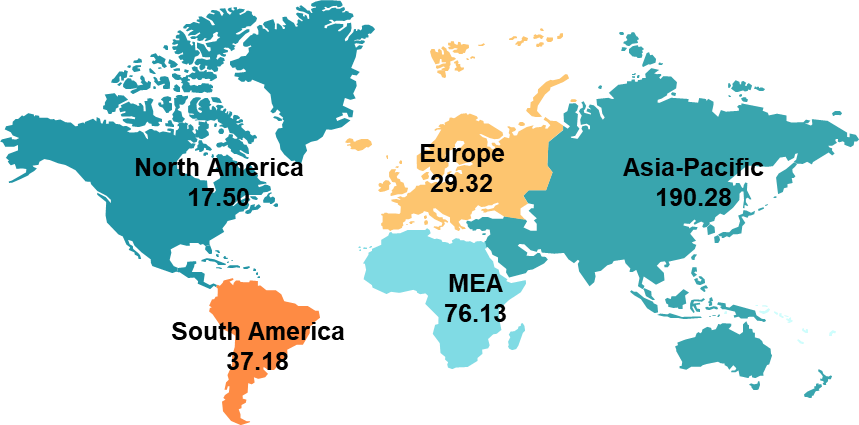
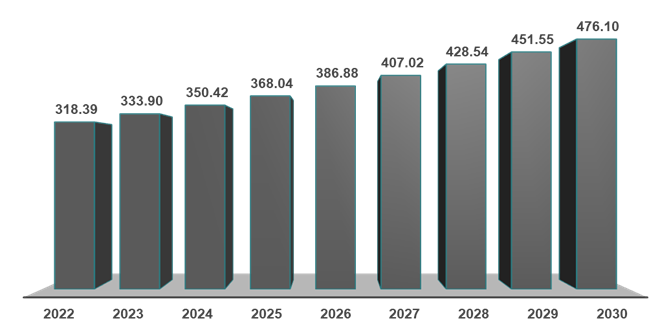
As per DataM intelligence estimates, nearly 371.4 million prevalent cases are estimated worldwide in 2025. The region with the highest prevalence is Asia-Pacific, accounting for 190.28 million cases. The prevalent cases of MASH across global regions are as follows:
As per DataM estimates, globally, the prevalent cases of MASH were estimated to be 368.04 million in 2025, 386.88 million in 2026, and a rise of up to 476.10 million by 2030.
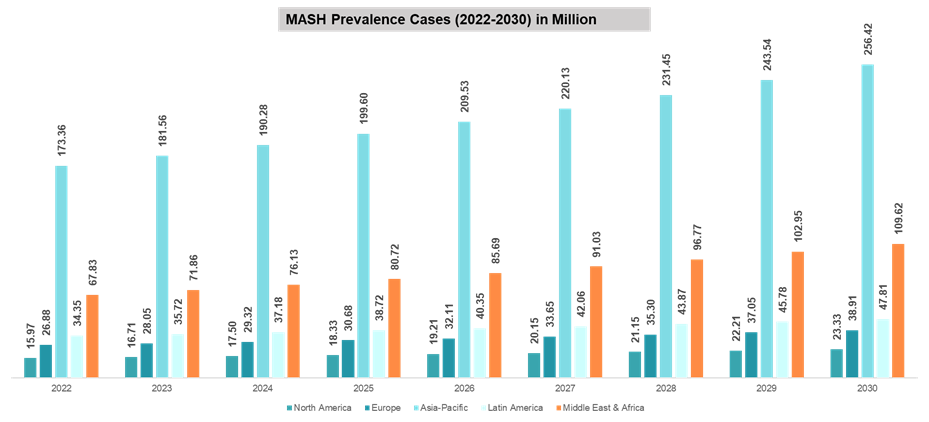
Epidemiology by Region:
Asia-Pacific is the leading region with the highest number of estimated MASH prevalent cases. In 2024, a total of up to 190.28 million prevalent cases were estimated in the region, which may reach up to 256.42 million by 2030. This higher prevalence is majorly attributed to the region’s huge population.
Global MASH Prevalent, Diagnosed and Treatment Population (2024) in Million
Globally, the overall prevalent cases of MASH in 2024 were 350.42 million, of which the diagnosed population were 37.84 million, and the patients who are taking one or the other treatment were 12.71 million.
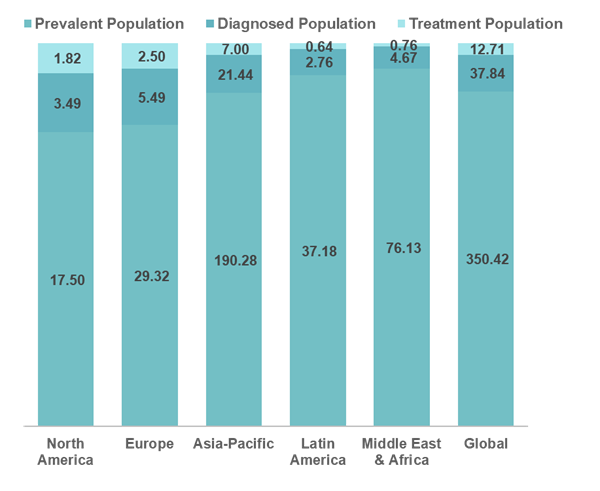
- In North America, the overall prevalent cases in 2024 were 17.50 million, of which the diagnosed population were estimated to be 3.49 million and among the diagnosed population, nearly 1.82 million were estimated to be getting treatment.
- In Europe, the overall prevalent cases in 2024 were 29.30 million, of which the diagnosed population were estimated to be 5.49 million and among the diagnosed population, nearly 2.50 million were estimated to be getting treatment.
- In Asia-Pacific, the overall prevalent cases in 2024 were 190.28 million, of which the diagnosed population were estimated to be 21.44 million and among the diagnosed population, nearly 7.00 million were estimated to be getting treatment.
- In Latin America, the overall prevalent cases in 2024 were 37.18 million, of which the diagnosed population was estimated to be 2.76 million, and among the diagnosed population, nearly 0.64 million were estimated to be getting treatment.
- In Middle East and Africa, the overall prevalent cases in 2024 were 76.13 million, of which the diagnosed population were estimated to be 4.67 million and among the diagnosed population, nearly 0.76 million were estimated to be getting treatment.
Recent Developments:
- February 2026: Multiple advanced pipeline drugs in the US advanced to Phase 3 trials, targeting anti-fibrotic, metabolic, and multi-target mechanisms to address liver histology and disease progression.
- January 2026: Madrigal Pharmaceuticals outlined an expansion strategy at JPM 2026, acquiring rights to Pfizer’s DGAT-2 inhibitor ervogastat and an oral GLP-1 agonist MGL-2086 to build early dominance in the emerging MASH market.
- December 2025: The MASH treatment market grew to $2.6 billion, driven by a 30% compound annual growth rate from rising prevalence of metabolic conditions, processed food diets, and sedentary lifestyles.