In-Silico Clinical Trials Market Size
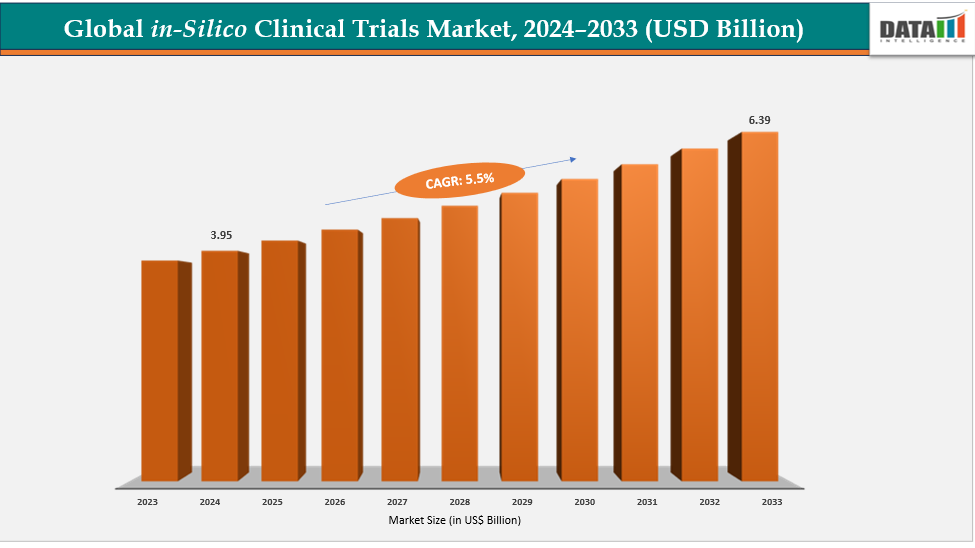
The global In-Silico Clinical Trials Market reached US$ 4.17 billion in 2025 and is expected to reach US$ 6.39 billion by 2033, growing at a CAGR of 5.5% during the forecast period 2026-2033.
The ability to rapidly replicate intricate biological and chemical processes is made possible by advances in computing power. To forecast the safety and effectiveness of drugs, AI algorithms examine vast datasets. Model accuracy is increased by expanding access to clinical and real-world data. Due to these variables, medication development is more rapid and less costly. Virtual trials help pharmaceutical companies cut expenses and time to market. It is advantageous for medtech companies to simulate device performance prior to human testing. Utilizing in-silico tools, contract research organizations (CROs) provide cutting-edge services. Investments in software and platforms are driven by strong commercial interest. Model-informed evidence is slowly being acknowledged by regulatory bodies.
In-Silico Clinical Trials Market Market Dynamics
Drivers: Increased research and development spending in pharma and biotech is significantly driving the in-Silico clinical trials market growth
The in-silico clinical trials market is driven by increased research and development (R&D) expenditures in biotechnology and pharmaceutical businesses. Companies are pushed to look for quicker and more affordable solutions by growing R&D budgets. Large-scale physical experiments are not always necessary thanks to in-silico trials. They reduce the risk to patients and save time and money. To forecast results and optimize drug candidates sooner, biotech companies are employing computer modeling. Investment in cutting-edge AI platforms and simulation tools is made possible by increased spending.
Restraints: Model credibility and validation requirements are hampering the growth of the in-Silico clinical trials market
The market expansion for in-silico clinical trials is being impeded by model believability and validation requirements. Before approving simulation-based results, regulatory bodies expect clear, dependable, and reproducible models. This necessitates thorough validation to guarantee that virtual models correctly forecast real-world events. This procedure is expensive, time-consuming, and technically challenging.
Furthermore, businesses face uncertainty due to the absence of globally recognized frameworks and disparate assessment standards among regulatory agencies. Additionally, many firms lack the know-how to carry out validation in accordance with stringent requirements.
For more details on this report, see Request for Sample
In-Silico Clinical Trials Market, Segment Analysis
The global in-Silico clinical trials market is segmented based on model type, application, clinical phase, end user and region.
The market for in-silico clinical trials is dominated by the pharmacokinetic (PK) and pharmacodynamic (PD) models. They are extensively employed in medication development. The movement of medicines through the body is studied by PK models. PD models examine the physiological effects of medicines. When combined, they aid in the prediction of dose-response relationships. PK/PD data is accepted by regulators for use in making decisions. They lessen the necessity of lengthy human testing. These models are established and thoroughly tested.
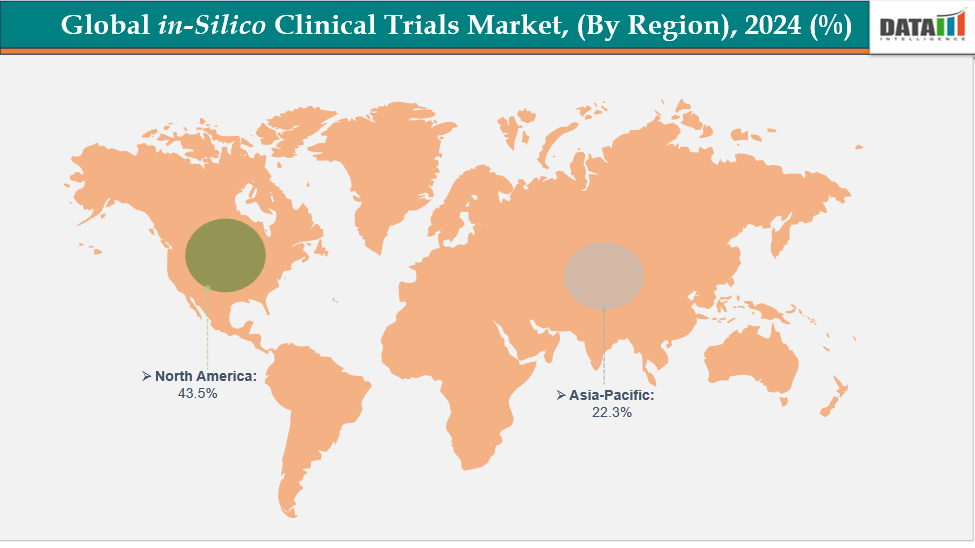
In-Silico Clinical Trials Market, Geographical Analysis
In-Silico Clinical Trials Market Competitive Landscape
Top companies in the in-Silico clinical trials market include Certara, Dassault Systèmes, InSilicoTrials Technologies, Nova, Insilico Medicine, The AnyLogic Company, Simulations Plus, VeriSIM Life, Physiomics Plc, and ANSYS, Inc., among others.
Certara: Certara is a global leader in biosimulation and AI-driven drug development, offering advanced modeling and simulation solutions to enhance clinical trial efficiency. Their platforms, such as Simcyp and Phoenix, enable virtual clinical trials, optimizing dosing, safety, and efficacy assessments. Trusted by over 2,600 companies worldwide, Certara's expertise accelerates regulatory submissions and supports informed decision-making across all stages of drug development
Recent Developments:
- April 2026 – Certara expands AI-driven biosimulation capabilities for virtual clinical development
Certara launched enhanced AI-enabled biosimulation and model-informed drug development tools to accelerate in-silico clinical trial design, dose optimization, and regulatory submission workflows for pharmaceutical companies. - April 2026 – Dassault Systèmes advances virtual twin technology for biomedical simulation
Dassault Systèmes expanded its Living Heart and virtual human modeling initiatives to support in-silico clinical trials, enabling pharmaceutical and medical device companies to simulate patient outcomes and improve trial efficiency. - March 2026 – Insilico Medicine advances AI-designed drug candidate into mid-stage clinical evaluation
Insilico Medicine announced continued progress of its AI-discovered fibrosis drug candidate with expanded clinical validation, highlighting the growing integration of AI modeling and in-silico simulations in drug discovery and trial optimization. - March 2026 – Simulations Plus strengthens PBPK and QSP simulation portfolio through software upgrades
Simulations Plus released major updates to its physiologically based pharmacokinetic (PBPK) and quantitative systems pharmacology (QSP) platforms to improve virtual patient modeling and predictive clinical trial simulation accuracy. - February 2026 – ANSYS expands digital twin simulation tools for medical device validation
ANSYS introduced advanced multiphysics simulation and digital twin technologies supporting in-silico testing for cardiovascular devices, implants, and personalized treatment planning in regulated healthcare environments. - January 2026 – InSilicoTrials Technologies collaborates with research institutions on virtual trial modeling
InSilicoTrials Technologies announced new collaborations focused on AI-enabled virtual patient cohorts and computational modeling platforms to support decentralized and simulation-based clinical trial strategies.
Market Scope
| Metrics | Details | |
| CAGR | 5.5% | |
| Market Size Available for Years | 2022-2033 | |
| Estimation Forecast Period | 2026-2033 | |
| Revenue Units | Value (US$ Bn) | |
| Segments Covered | By Model Type | Pharmacokinetic and pharmacodynamic Models, Physiologically Based PK, Quantitative Systems Pharmacology, Virtual Patient and Digital Twin, Computational Fluid Dynamics and Finite Element Models and Others |
| By Application | Drug Development, Medical Device Evaluation, Regulatory Submissions, Post-Market Surveillance and Others | |
| By End User | Pharmaceutical and Biotech Companies, Medical Device Manufacturers, Academic and Research Institutes, contract research organizations, and Others | |
| Regions Covered | North America, Europe, Asia-Pacific, South America and the Middle East & Africa | |
The global in-Silico clinical trials market report delivers a detailed analysis with 62 key tables, more than 57 visually impactful figures, and 159 pages of expert insights, providing a complete view of the market landscape.
Suggestions for Related Report
For more pharmaceuticals-related reports, please click here