Chronic Spontaneous Urticaria (SCU) Treatment Market: Industry Outlook
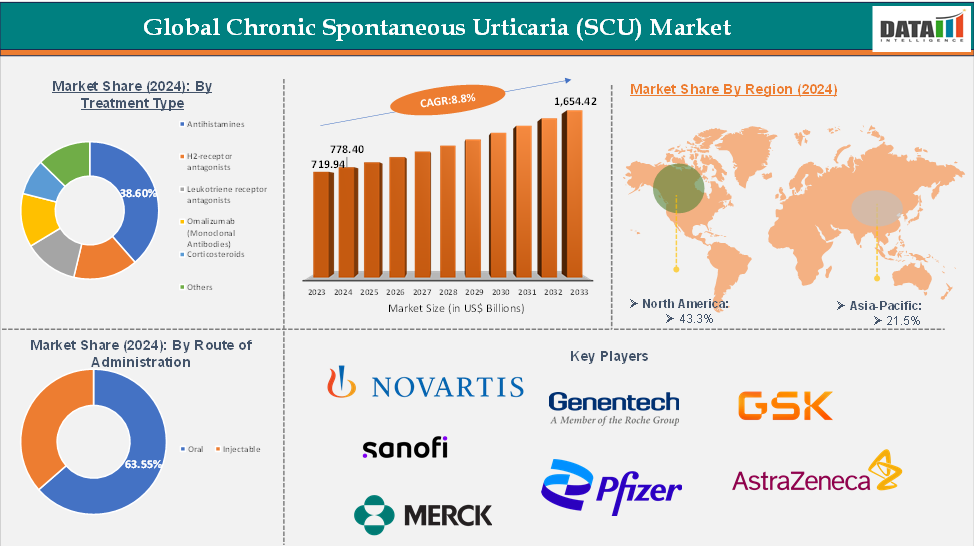
nbsp;1,654.42 Million by 2033, growing at a CAGR of 8.8% during the forecast period 2025-2033.The Global Chronic Spontaneous Urticaria (SCU) Treatment Market reached US$ 719.94 Million in 2023, with a rise of US$ 778.40 Million in 2024 and is expected to reach US
The chronic spontaneous urticaria (CSU) market is experiencing significant growth due to a surge in dermatological and immunological disorders affecting millions globally. CSU, also known as chronic idiopathic urticaria, is characterized by the spontaneous appearance of hives and/or angioedema lasting for six weeks or more without a known cause. As awareness and diagnosis improve, the demand for treatment is rising across both developed and developing nations.
Advancements in biologic drugs, increased healthcare access, and innovation in oral antihistamines and immune-modulating therapies are supporting this growth. The market's future trajectory is shaped by growing R&D investments, expanding reimbursement frameworks in high-income countries, and patient-centric approaches in dermatology.
Chronic Spontaneous Urticaria (SCU) Treatment Market: Executive Summary
Global Chronic Spontaneous Urticaria (SCU) Treatment Market Dynamics: Drivers & Restraints
Driver: Rising prevalence of autoimmune and allergic skin conditions
The CSU market is expanding due to the rise in autoimmune and allergic skin diseases, often coexisting with other conditions like systemic lupus erythematosus, autoimmune thyroid disease, or rheumatoid arthritis. Lifestyle factors like pollution, allergen exposure, diet changes, and stress have also contributed to increased allergic reactions and hypersensitivity disorders.
As more people seek medical consultation for recurrent hives, diagnosis rates have improved, leading to a growing demand for effective therapies. The chronic nature of CSU necessitates long-term management plans, requiring sustained pharmaceutical intervention. As awareness grows, the market is expected to grow in urban and semi-urban regions.
For instance, autoimmunity is often linked to developed Western countries, with higher incidence rates in industrial regions like Northern Europe and North America. A study found that autoimmune diseases affect 5-10% of the industrialized world population, while developing countries have a lower prevalence but are on the rise.
Driver: Advancements in biologic therapies
Biologic therapies have significantly impacted the treatment of Chronic Substance Use Disorder (CSU), particularly in patients who are resistant to standard antihistamines. The introduction of omalizumab, a monoclonal anti-IgE antibody, has revolutionized the management of CSU, offering improved symptom control, reduced flare-ups, and improved quality of life with fewer side effects.
As the global pharmaceutical industry invests in immunological drug pipelines, more biologic candidates are entering clinical trials. The inclusion of omalizumab in dermatology guidelines has boosted its adoption. Technological advancements, enhanced delivery systems, and personalized treatment regimens are also playing crucial roles in expanding this segment, positioning biologics as a cornerstone in future CSU management.
Restraint: High cost of advanced therapies
The high cost of biologic drugs like omalizumab, which are effective in treating chronic subcutaneous uterine disease (CSU), is a major barrier to market growth. These drugs are particularly expensive in middle- and low-income countries where insurance coverage is limited or non-existent. Patients often have to bear out-of-pocket expenses, making long-term treatment financially unsustainable.
Even in developed countries, budget constraints and strict reimbursement policies restrict patient access. The chronic nature of CSU often requires continuous therapy, further compounding the cost burden. The lack of generic or biosimilar alternatives further exacerbates the issue. Without a shift in pricing models, policy support, or affordable alternatives, the high cost will continue to hinder the global expansion of advanced CSU therapies.
Opportunity: Emergence of novel small-molecule therapies and personalized medicine
The personalized medicine and small molecule development field offers a significant opportunity in the cosmetic surgery (CSU) market. As researchers and pharmaceutical companies understand the molecular and immunological underpinnings of CSU, they can identify novel targets for intervention. Small-molecule therapies offer oral administration, better patient compliance, and lower manufacturing costs.
Advancements in genomics and biomarker discovery allow clinicians to tailor treatment strategies based on individual patient profiles, improving efficacy and reducing trial-and-error prescribing. The integration of artificial intelligence and machine learning in clinical decision-making helps identify treatment responders early. As regulatory agencies support fast-track approvals for innovative treatments, the pipeline of small-molecule and personalized therapies is expected to become a major growth driver over the next decade.
For more details on this report, Request for Sample
Global Chronic Spontaneous Urticaria (SCU) Treatment Market Segment Analysis
The global Chronic Spontaneous Urticaria (SCU) Treatment Market is segmented based on treatment type, route of administration, distribution channel, and region.
Treatment Type:
The antihistamines segment from the treatment type is expected to hold 38.60% of the chronic spontaneous urticaria (SCU) market
Antihistamines are a key component of chronic syncytial urticaria (CSU) treatment due to their accessibility, affordability, and efficacy. Both first-generation and second-generation options are prescribed to manage symptoms like itching, redness, and swelling. Second-generation options like loratadine, cetirizine, and fexofenadine are preferred due to their non-sedating properties and longer duration of action.
The widespread over-the-counter availability of these drugs boosts market penetration, especially in regions with limited healthcare access. Physicians often recommend up-dosing for moderate symptoms, contributing to volume sales. Strong generic competition ensures affordability and high market visibility, making it a foundational segment in CSU therapy.
For instance, in April 2025, the FDA approved Dupixent for treating adults and adolescents aged 12 and older with chronic spontaneous urticaria who continue to show symptoms despite histamine-1 antihistamine treatment.
Global Chronic Spontaneous Urticaria (SCU) Treatment Market - Geographical Analysis
North America is the global Chronic Spontaneous Urticaria (SCU) Treatment Market with a market share of 43.3% in 2024
North America dominates the Cosmetic Substances (CSU) market due to its mature healthcare ecosystem, high awareness, and rapid adoption of advanced treatment modalities. The US has a well-developed clinical infrastructure, global pharmaceutical leaders, and widespread access to biologics and immunotherapies.
Insurance coverage and reimbursement support for costly treatments boost patient adherence. High investments in biomedical research and clinical trials support innovation. Leading academic institutions and healthcare providers contribute to clinical guidelines, encouraging evidence-based practices in CSU management. A proactive approach to physician education, patient advocacy, and specialty care coordination ensures timely diagnosis and effective long-term disease management.
For instance, in January 2025, Allakos announced a 75% workforce reduction and discontinuation of its CSU AK006 development programme due to disappointing Phase I results, leaving the US biotech with around 15 employees while evaluating strategic alternatives.
Asia-Pacific is the global Chronic Spontaneous Urticaria (SCU) Treatment Market with a market share of 21.5% in 2024
The Asia-Pacific region is gaining momentum as a growth market for CSU treatment due to factors such as urbanization, lifestyle changes, and environmental pollution. Rising disposable incomes, awareness about skin health, and expanding access to dermatological care are improving the diagnosis and treatment of CSU.
Moreover, regional governments are investing in healthcare infrastructure, and specialized allergy and dermatology clinics are growing. Pharmaceutical companies are shifting their focus to Asia due to favorable regulatory environments, lower clinical trial costs, and large patient pools. Cost-effective treatment options, such as generics and local manufacturing capabilities, are also contributing to market penetration.
For instance, in February 2024, Japan's Ministry of Health, Labor and Welfare approved Dupixent (dupilumab) for treating chronic spontaneous urticaria in individuals aged 12 and older who cannot be adequately controlled with existing therapies. This marks Japan's first approval for CSU, emphasizing Dupixent's value as a novel treatment option for unmet needs.
Global Chronic Spontaneous Urticaria (SCU) Treatment Market – Major Players
The major players in the Chronic Spontaneous Urticaria (SCU) Treatment Market include Novartis AG, Genentech, Inc. (Roche Group), GlaxoSmithKline plc, Sanofi S.A., Pfizer Inc., AstraZeneca, Merck & Co., Inc., Teva Pharmaceuticals, Mylan N.V., and Sun Pharmaceutical Industries Ltd., among others.
Global Chronic Spontaneous Urticaria (SCU) Treatment Market – Key Developments
In March 2025, the Phase III REMIX-1 and REMIX-2 clinical trials reveal that remibrutinib effectively reduces chronic spontaneous urticaria symptoms in patients unresponsive to second-generation H1-antihistamines. The drug's rapid onset, sustained efficacy, and favorable safety profile were found in the study published by The New England Journal of Medicine.
Chronic Spontaneous Urticaria (SCU) Treatment Market: Scope
Metrics | Details | |
CAGR | 8.8% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Mn) | |
Segments Covered | Treatment Type | H2-receptor antagonists, Leukotriene receptor antagonists, Omalizumab (Monoclonal Antibodies), Corticosteroids, Others |
Route of Administration | Oral, Injectable | |
Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
DMI Insights:
The global chronic spontaneous urticaria (CSU) market, valued at $719.94 million in 2023, is projected to reach $1,654.42 million by 2033, growing at an 8.8% CAGR.
The market is driven by increasing awareness of autoimmune and allergic skin conditions, improved diagnostics, and demand for long-term therapies. Advancements in biologic treatments and novel approvals are transforming clinical practice, but affordability and access remain challenges.
North America leads the market due to robust healthcare infrastructure and biologic adoption, while Asia-Pacific shows promising growth due to improved access and regulatory support. Future opportunities lie in small-molecule innovation, personalized medicine, and AI-driven clinical decision-making.
The global Chronic Spontaneous Urticaria (SCU) Treatment Market report delivers a detailed analysis with 62 key tables, more than 54 visually impactful figures, and 185 pages of expert insights, providing a complete view of the market landscape.
Suggestions for Related Report
For more pharmaceuticals-related reports, please click here