Central America Certified Reference Materials Market Overview
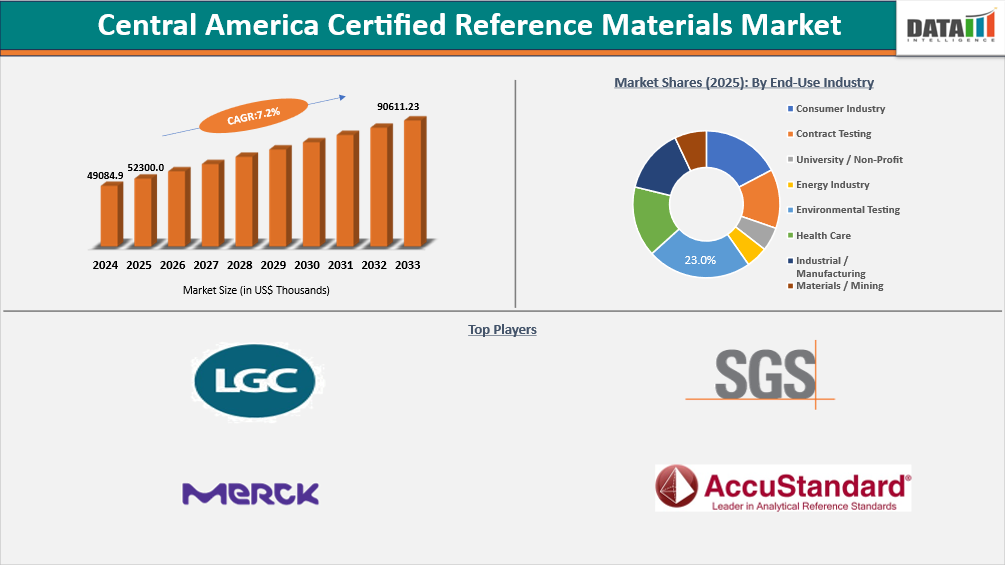
Central America Certified Reference Materials Market reached US$ 52,300.00 Thousands in 2025 and is expected to reach up to US$ 90,611.23 Thousands in 2033, growing at a CAGR of 7.2% during the forecasting period (2026-2033). The Central America certified reference materials (CRM) market is growing alongside the region’s expanding analytical testing infrastructure across mining, environmental monitoring, pharmaceuticals, and food safety. Laboratories rely on CRMs for equipment calibration, method validation, and traceability to meet ISO/IEC 17025 accreditation requirements, with demand rising as regulatory frameworks and trade activities strengthen. The mining sector in countries such as Panama, Nicaragua, and Guatemala is a key driver, creating steady demand for inorganic and geological CRMs used in high-precision techniques like XRF and ICP-MS.
Additionally, regional harmonization efforts through SIECA and increasing environmental regulations are accelerating CRM adoption in water and soil monitoring. As governments focus on pollution control and biodiversity protection, laboratories require reliable reference materials for accurate contaminant analysis. Overall, growing industrial activity and regulatory enforcement are positioning CRMs as essential tools for quality assurance and compliance across Central America.
Certified Reference Materials Industry Trends and Strategic Insights
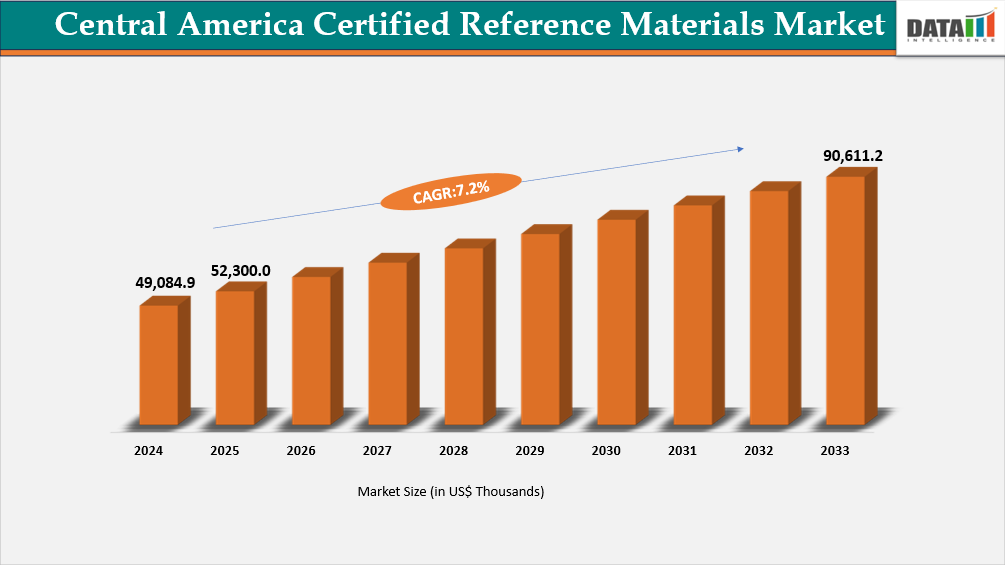
- By end-user industry, the environmental testing is projected to be the largest market, holding a significant share of about 25.2% in 2025.
Central America Certified Reference Materials Market Size and Future Outlook
- 2025 Market Size: US$ 52,300.0 Thousands
- 2033 Projected Market Size: US$ 90,611.23 Thousands
- CAGR (2026-2033): 7.2%
Market Scope
| Metrics | Details |
| By Product Type | Organic, Inorganic |
| By Application | Quality Control & Assurance, Method Validation, Regulatory Compliance Testing, R&D & Analytical Development, Calibration & Instrument Validation |
| By End-User Industry | Consumer Industry, Contract Testing, University / Non-Profit, Energy Industry, Environmental Testing, Health Care, Industrial / Manufacturing, Materials / Mining |
| By Country | Panamá, Costa Rica, Guatemala, El Salvador, Honduras, Others |
| Report Insights Covered | Competitive Landscape Analysis, Company Profile Analysis, Market Size, Share, Growth |
Market Dynamics
Expanding Pharmaceutical Manufacturing Drives Demand For Accurate Analytical Calibration Standards
The rapid expansion of pharmaceutical manufacturing across Central America is a key driver of the certified reference materials (CRM) market. Countries such as Costa Rica, Guatemala, and El Salvador are developing strong pharmaceutical ecosystems, attracting multinational companies and boosting local generic drug production. Costa Rica, in particular, has emerged as a regional hub due to favorable trade agreements, skilled workforce availability, and supportive government policies, increasing the need for high-quality analytical standards.
As pharmaceutical production scales, CRMs have become essential for ensuring regulatory compliance across drug development, quality control, and post-market surveillance. Applications such as API quantification, impurity profiling, and stability testing rely on traceable, ISO 17034-certified reference materials. Additionally, stricter enforcement of Good Manufacturing Practice (GMP) standards across countries like Panama and Honduras is further driving demand for precise calibration standards, reinforcing CRM adoption in the regional pharmaceutical sector.
High Production and Certification Costs Limiting Adoption Among Smaller Laboratories
The high cost structure of certified reference materials (CRMs) remains a significant restraint in the Central America market, particularly for small and medium-sized laboratories. Limited domestic production forces heavy reliance on imports, leading to additional expenses from duties, logistics, and currency fluctuations, often increasing prices by 20–35%. This creates financial pressure on laboratories in countries such as Honduras, Nicaragua, and Guatemala, where budget constraints make it difficult to balance regulatory compliance with operational costs.
Additionally, the complex and expensive process of producing ISO 17034-certified CRMs, especially ultra-high-purity materials required for pharmaceutical applications, further elevates prices. These cost barriers limit accessibility for smaller laboratories and reduce overall CRM adoption rates. Without supportive measures such as regional subsidies, pooled procurement, or government-backed initiatives, affordability challenges are likely to continue hindering market growth.
Segmentation Analysis
The Central America certified reference materials market is segmented based on product type, application, end-user industry, and region.
Environmental Regulations and Climate Commitments Drive Environmental Testing in the Central America CRM Market
Environmental testing is the dominant segment in the Central America certified reference materials (CRM) market, driven by expanding regulatory requirements across water, soil, air, and ecological monitoring. Laboratories rely on metrologically traceable CRMs to ensure accuracy and compliance in applications such as water quality testing, waste analysis, and biodiversity assessments. Strengthening environmental governance, supported by international trade obligations and climate commitments, is significantly increasing the demand for reliable reference standards across the region.
Water quality monitoring represents the largest application area, with countries aligning to global benchmarks such as the U.S. EPA Water Quality Standards, requiring testing of numerous pollutants using CRM-calibrated methods. Additionally, regional commitments toward net-zero emissions and the development of climate monitoring frameworks are driving demand for gas standards and environmental matrix CRMs. This combination of regulatory enforcement and sustainability initiatives firmly establishes environmental testing as the leading segment in the Central America CRM market.
Regulatory Strengthening and Medical Device Expansion Drive Healthcare CRM Demand
Healthcare is a key and high-value segment in the Central America certified reference materials (CRM) market, driven by increasing reliance on accurate and traceable analytical results across clinical diagnostics, pharmaceutical quality control, and medical device testing. Laboratories, hospital pharmacies, and public health institutions depend on CRMs to ensure precision in patient safety decisions, particularly as regional healthcare systems adopt stricter regulatory frameworks and international accreditation standards such as ISO 15189.
Regulatory harmonization efforts led by the Pan American Health Organization (PAHO), in collaboration with the U.S. Food and Drug Administration (FDA), are accelerating the adoption of GMP and GLP standards, which mandate the use of certified reference materials. The growing presence of global medical device manufacturers in Costa Rica further strengthens demand for high-quality CRMs, positioning healthcare as one of the most strategically important and rapidly evolving segments in the region.
Geographical Penetration
Panama Certified Reference Materials Market Trends
Panama’s certified reference materials (CRM) market is supported by a strong national quality infrastructure led by Comisión Panameña de Normas Industriales y Técnicas (COPANIT) under Ministerio de Comercio e Industrias (MICI), which enforces ISO-aligned metrology and accreditation standards. The presence of advanced laboratories such as Intertek Panama, operating with ISO/IEC 17025 accreditation, highlights the country’s growing analytical capabilities and reliance on high-purity CRMs for calibration and validation across petroleum, environmental, and food testing applications.
Environmental monitoring plays a critical role, particularly in water quality testing linked to the Panama Canal’s freshwater systems, where accurate chemical analysis has become a strategic priority. Additionally, increasing ISO/IEC 17025 laboratory accreditation, growth in pharmaceutical and medical device activities, and alignment with international trade frameworks such as CAFTA-DR and WTO standards are driving sustained CRM demand. These factors collectively position Panama as a key and steadily growing market within Central America
Costa Rica Certified Reference Materials Market Outlook
Costa Rica’s certified reference materials (CRM) market is uniquely driven by its advanced life sciences sector, which includes a strong presence of pharmaceutical and medical device companies. With over 90 MedTech multinationals and leading global pharmaceutical firms operating in the country, there is a high and consistent demand for CRMs to support calibration, validation, and regulatory compliance across manufacturing and testing activities. Institutions such as the National Center for Biotechnological Innovation (CENIBiot) and research initiatives by Pfizer further strengthen CRM demand through R&D and clinical research applications.
The country’s Free Trade Zone (FTZ) regime, supported by Promotora del Comercio Exterior (PROCOMER), provides tax and import duty benefits that significantly reduce CRM procurement costs compared to other Central American markets. This favorable economic environment, combined with ISO/IEC 17025-aligned quality systems and growing pharmaceutical innovation, positions Costa Rica as a high-value and rapidly expanding CRM market in the region.
Product and Portfolio Analysis
Stock Vs Custom Capabilities
Central America is witnessing a steady rise in ISO/IEC 17025-accredited laboratories, particularly in Costa Rica, Panama, and Guatemala, driven by growth in pharmaceuticals, food exports, and environmental monitoring. The region’s compliance-focused laboratory base relies heavily on stock CRMs for routine calibration and regulatory testing, ensuring adherence to international trade standards. Strong agricultural exports and alignment with Codex and US/EU regulations further reinforce demand for ready-to-use standards, especially for pesticide residue and contaminant testing.
Environmental and public health monitoring is another key driver, with increasing focus on water quality and pollutant control across countries like Panama and Costa Rica. Laboratories depend on standardized inorganic CRMs for testing heavy metals such as lead, arsenic, and mercury in water systems. The market remains largely stock-driven, with limited adoption of custom CRMs, except in niche areas such as pharmaceutical formulation testing and specialized environmental studies. Global suppliers like Merck KGaA, LGC Limited, and AccuStandard dominate supply through regional distributors, ensuring timely availability of CRM solutions.
Customer Buying Behavior Analysis
Switching Triggers
Supplier switching in the Central America CRM market is primarily driven by supply chain reliability and local availability constraints, as laboratories depend heavily on a limited distributor network. Import delays, customs challenges, and inventory gaps can disrupt routine testing workflows, prompting laboratories to shift toward suppliers with regional hubs, stronger in-country stocking, and faster delivery timelines. Vendors offering consistent availability and shorter turnaround times gain a clear competitive advantage.
Additionally, evolving regulatory requirements and technical expectations play a critical role in switching decisions. The introduction of new testing programs, stricter ISO/IEC 17025 compliance, and increasing export standards drive demand for broader CRM portfolios and robust documentation. Laboratories also prioritize suppliers with consistent product quality, strong certification traceability, and responsive technical support, especially as advanced analytical techniques become more prevalent.
Regulatory Analysis
Regulatory frameworks across Central America are a primary driver of certified reference materials (CRM) demand, particularly in food, pharmaceutical, environmental, and agricultural sectors. Regional standards such as RTCA 67.01.31:20 for processed foods and RTCA 11.03.59:18 for pharmaceuticals mandate strict testing for contaminants, additives, and GMP compliance, increasing the need for pesticide CRMs, food matrix standards, and API reference materials. Additionally, Panama’s food safety regulations and regional pesticide/fertilizer frameworks are reinforcing demand for GMO standards and high-purity pesticide CRMs to support export-oriented agriculture.
Environmental and trade-related regulations further strengthen CRM adoption across the region. Costa Rica’s MINAE standards require monitoring of water quality, heavy metals, and hazardous substances, while CAFTA-DR compliance enforces alignment with US FDA/EPA standards, making traceable analytical results essential for international trade. Across all countries, ISO/IEC 17025 accreditation mandates strict traceability and method validation, creating continuous baseline demand for CRMs. Emerging environmental policies and increasing enforcement of pollutant monitoring, including PFAS and industrial runoff, are further accelerating the need for specialized and ultra-trace reference materials.
Competitive Landscape
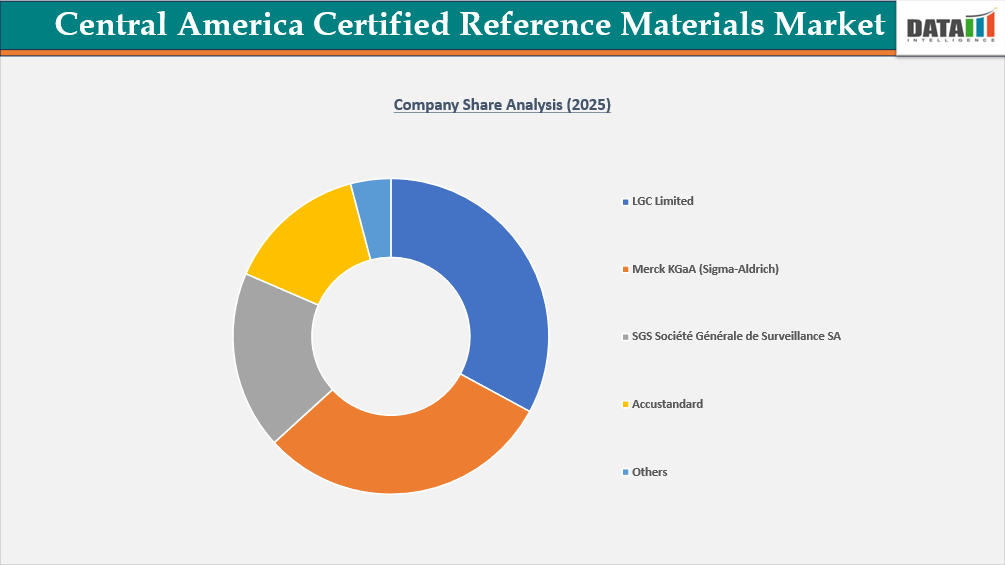
- The Central America certified reference materials market is characterized by a competitive landscape that includes both established and regional players.
- Key players include LGC Limited, Merck KGaA, SGS Société Générale de Surveillance SA, and AccuStandard
Why Choose DataM?
- Data-Driven Insights: Dive into detailed analyses with granular insights such as pricing, market shares, and value chain evaluations, enriched by interviews with industry leaders and disruptors.
- Post-Purchase Support and Expert Analyst Consultations: As a valued client, gain direct access to our expert analysts for personalized advice and strategic guidance, tailored to your specific needs and challenges.
- White Papers and Case Studies: Benefit quarterly from our in-depth studies related to your purchased titles, tailored to refine your operational and marketing strategies for maximum impact.
- Annual Updates on Purchased Reports: As an existing customer, you enjoy the privilege of annual updates to your reports, ensuring you stay abreast of the latest market insights and technological advancements. Terms and conditions apply.
- Specialized Focus on Emerging Markets: DataM differentiates itself by delivering in-depth, specialized insights specifically for emerging markets, rather than offering generalized geographic overviews. This approach equips our clients with a nuanced understanding and actionable intelligence that are essential for navigating and succeeding in high-growth regions.
- Value of DataM Reports: Our reports offer specialized insights tailored to the latest trends and specific business inquiries. This personalized approach provides a deeper, strategic perspective, ensuring you receive the precise information necessary to make informed decisions. These insights complement and go beyond what is typically available in generic databases.
Target Audience 2026
- Manufacturers/ Buyers
- Industry Investors/Investment Bankers
- Research Professionals
- Emerging Companies
























































