Alpha-1 Antitrypsin (AAT) Deficiency Market Size
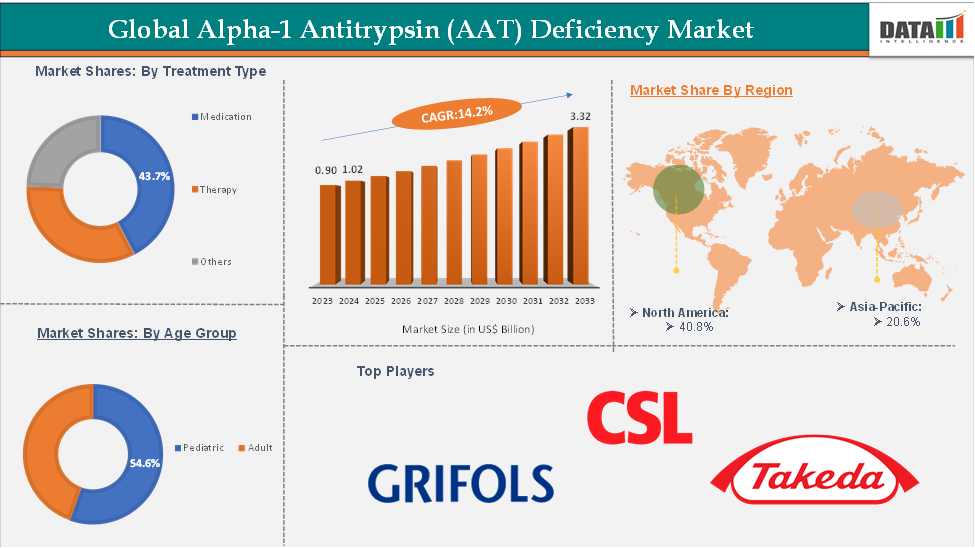
The global alpha-1 antitrypsin (AAT) deficiency was valued at US$ 0.90 Billion in 2023. The global Alpha-1 Antitrypsin (AAT) Deficiency market size reached US$ 1.02 Billion in 2024 and is expected to reach US$ 3.32 Billion by 2033, growing at a CAGR of 14.2% during the forecast period 2025-2033.
Alpha-1 Antitrypsin (AAT) Deficiency Market Overview
The alpha-1 antitrypsin (AAT) deficiency market is experiencing steady growth driven by increasing disease prevalence, improved diagnostic capabilities, and rising awareness among both patients and healthcare providers. The market is further supported by a robust pipeline of innovative therapies, including gene and RNA-based treatments, and favorable regulatory incentives such as orphan drug designation and fast-track approvals.
The growing focus on biosimilars, cost-effective alternatives, and expanded newborn screening programs presents strong opportunities for future expansion.
North America currently leads the market due to its advanced healthcare infrastructure, robust reimbursement systems, and early access to novel treatments. In contrast, the Asia-Pacific region is projected to grow at the fastest pace, driven by increasing healthcare investment, rising awareness, and the gradual adoption of rare disease diagnostics across the region.
Alpha-1 Antitrypsin (AAT) Deficiency Market Executive Summary
Alpha-1 Antitrypsin (AAT) Deficiency Market Dynamics: Drivers & Restraints
Rising prevalence & improved awareness are significantly driving the alpha-1 antitrypsin (AAT) deficiency market growth
The increasing prevalence of alpha-1 antitrypsin (AAT) deficiency, along with growing awareness among patients and healthcare providers, is playing a key role in driving the market forward. In the past, many cases went undiagnosed due to a limited understanding of the condition and its symptoms. For instance, according to the National Institute of Health in 2024, Alpha-1 Antitrypsin (AAT) Deficiency is a rare genetic disorder affecting approximately 1 in 2,000 to 6,000 people, increasing the risk of liver disease and early-onset emphysema. It is more commonly observed in individuals of European descent and is less prevalent among Asian and other populations.
Although classified as rare, the condition is significantly underdiagnosed. An estimated 80,000 to 100,000 people in the United States are believed to have severe AAT deficiency. Globally, over 3 million individuals are estimated to carry genetic variants linked to severe forms of the disorder. However, with improved access to information, targeted awareness campaigns, and support from patient advocacy groups, more individuals are now being identified and diagnosed earlier. This has led to a higher number of people seeking medical care and treatment for AAT deficiency.
As a result, the demand for effective therapies is increasing, encouraging further investment and development within the market..
Robust pipeline & regulatory incentives are significantly driving the alpha-1 antitrypsin (AAT) deficiency market growth
A robust pipeline of innovative therapies, supported by favorable regulatory incentives, is one of the major forces driving growth in the Alpha-1 Antitrypsin (AAT) deficiency market. Several pharmaceutical and biotech companies are actively developing advanced treatments, including gene therapy, RNA-based therapies, and inhalable formulations, aimed at addressing both the root cause and symptoms of the condition.
For instance, in March 2025, Beam Therapeutics Inc. reported initial safety and efficacy results from its Phase 1/2 trial of BEAM-302, demonstrating clinical proof-of-concept for treating Alpha-1 Antitrypsin Deficiency (AATD) using in vivo base editing. Many of these candidates are progressing through early- and mid-stage clinical trials, showing promising results.
At the same time, regulatory agencies such as the FDA and EMA are offering incentives like orphan drug designation, fast track status, and extended market exclusivity. These programs not only encourage research and development by reducing regulatory hurdles but also make it more financially viable for companies to invest in therapies for rare diseases like AAT deficiency.
Together, the expanding pipeline and supportive regulatory environment are expected to accelerate the availability of more effective, convenient, and long-term treatment options for patients, further strengthening market growth.
Restraint:
High cost of therapy hampering the growth of the Alpha-1 Antitrypsin (AAT) Deficiency market
The high cost of therapy remains a major barrier to the growth of the Alpha-1 Antitrypsin (AAT) deficiency market. For example, according to Grifols’ annual report and industry data, Prolastin-C can cost around $90,000 to $127,000 per patient annually in the U.S. This price reflects weekly lifelong infusions and associated healthcare services.
Such a high financial burden not only limits accessibility for patients without comprehensive insurance but also places pressure on national healthcare systems and insurers, often resulting in restricted reimbursement policies.
Opportunity:
Biosimilars & cost-effective alternatives are expected to create a lucrative opportunity for the growth of the Alpha-1 Antitrypsin (AAT) Deficiency market
The development of biosimilars and cost-effective alternatives presents a strong opportunity for growth in the Alpha-1 Antitrypsin (AAT) deficiency market. With current augmentation therapies being extremely expensive, the entry of biosimilars can help reduce treatment costs and improve accessibility for a larger patient population.
As patents for branded products like Prolastin-C and Zemaira approach expiration in key markets, there is room for manufacturers to introduce lower-cost alternatives without compromising efficacy or safety. This shift can be especially beneficial in regions where reimbursement systems are limited or out-of-pocket costs are high.
For more details on this report – Request for Sample
Alpha-1 Antitrypsin (AAT) Deficiency Market, Segment Analysis
The global alpha-1 antitrypsin (AAT) deficiency market is segmented based on disease type, treatment type, route of administration, and region.
The alpha-1 proteinase inhibitor from the medication in the treatment type segment are expected to hold 43.7% of the market share in 2024 in the alpha-1 antitrypsin (AAT) deficiency market
The alpha-1 proteinase inhibitor segment is expected to dominate the alpha-1 antitrypsin (AAT) deficiency market due to its status as the only disease-specific treatment currently approved for slowing disease progression. These inhibitors, used in augmentation therapy, directly address the underlying protein deficiency that causes lung damage in AATD patients, making them the cornerstone of clinical management. Their effectiveness in preserving lung function, reducing exacerbations, and improving quality of life has made them the most widely adopted therapeutic option.
Moreover, strong regulatory support, including orphan drug designation and reimbursement coverage in developed markets like the U.S. and Europe, has further reinforced their widespread use. For instance, in March 2025, Takeda announced that the U.S. Food and Drug Administration (FDA) had approved an update to the U.S. Prescribing Information for GLASSIA (Alpha-1 Proteinase Inhibitor [Human]), now including newly available 1-, 4-, and 5-gram single-dose vial options.
The availability of multiple branded products such as Prolastin-C, Zemaira, and Aralast has created a competitive landscape, improving supply and accessibility. In addition, the rising prevalence of diagnosed cases due to improved awareness and screening is expanding the eligible patient pool.
Alpha-1 Antitrypsin (AAT) Deficiency Market, Geographical Analysis
North America is expected to dominate the global alpha-1 antitrypsin (AAT) deficiency market with a 40.8% share in 2024
North America is expected to dominate the Alpha-1 Antitrypsin (AAT) deficiency market due to a combination of favorable healthcare infrastructure, higher diagnosis rates, and strong regulatory and reimbursement frameworks. The United States, in particular, has a well-established system for rare disease management, including access to advanced diagnostic tools like genetic screening and pulmonary function testing, which significantly improves early detection and patient identification.
Furthermore, the region benefits from a high level of awareness among clinicians, supported by ongoing educational efforts and robust patient advocacy through organizations like the Alpha-1 Foundation.
Additionally, most of the leading pharmaceutical companies developing and commercializing AAT therapies are either based in or have strong operations in North America, allowing for easier regulatory navigation and quicker market access. For instance, in June 2023, Arrowhead Pharmaceuticals Inc. announced updated data from its Phase 2 SEQUOIA clinical trial evaluating fazirsiran (TAK-999/ARO-AAT), an investigational therapy for liver disease linked to alpha-1 antitrypsin deficiency (AATD-LD).
Regulatory agencies like the FDA have granted orphan drug designations, fast-track status, and other incentives that help speed up the development and approval of novel treatments in this region. The presence of comprehensive insurance systems and government-supported reimbursement for high-cost therapies, especially under Medicare and private plans, also enhances therapy adoption.
Asia-Pacific is growing at the fastest pace in the alpha-1 antitrypsin (AAT) deficiency market, holding 20.6% of the market share
Asia-Pacific is seeing the fastest growth in the Alpha-1 Antitrypsin (AAT) Deficiency market, driven by a mix of rising disease prevalence and evolving lifestyles. With more people experiencing autoimmune conditions like Alpha-1 Antitrypsin (AAT) Deficiency, the demand for effective treatment is rising. Rapid urbanization and better awareness of skin and hair health are prompting more individuals to seek medical help.
At the same time, countries like China, Japan, South Korea, and India are ramping up investments in healthcare systems, dermatology research, and advanced treatment options, creating strong momentum for market expansion across the region.
Alpha-1 Antitrypsin (AAT) Deficiency Market Competitive Landscape
Top companies in the alpha-1 antitrypsin (AAT) deficiency market include Grifols, CSL Behring LLC, and Takeda Pharmaceutical Company Limited, among others.
Alpha-1 Antitrypsin (AAT) Deficiency Market Key Developments
In July 2024, Intellia Therapeutics, Inc. announced the authorization of its Clinical Trial Application (CTA) by the United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA) to initiate a Phase 1/2 study evaluating NTLA-3001 for the treatment of alpha-1 antitrypsin deficiency (AATD)-associated lung disease.
Alpha-1 Antitrypsin (AAT) Deficiency Market Scope
Metrics | Details | |
CAGR | 7.5% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Bn) | |
Segments Covered | Treatment Type | Medication, Therapy, Others |
Age Group | Pediatric, Adult | |
Route of Administration | Parenteral, Intranasal, Others | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
The global alpha-1 antitrypsin (AAT) deficiency market report delivers a detailed analysis with 60+ key tables, more than 55+ visually impactful figures, and 178 pages of expert insights, providing a complete view of the market landscape.
Suggestions for Related Report
For more pharmaceutical-related reports, please click here