Scleroderma Market Size
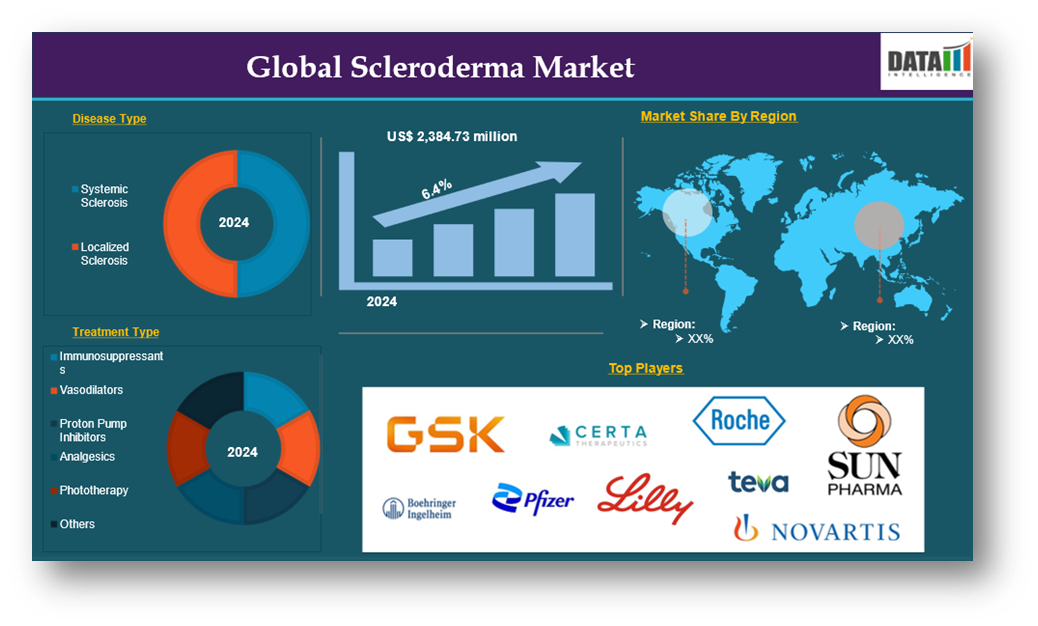
The global scleroderma market reached US$ 2,384.73 million in 2024 and is expected to reach US$ 4,103.07 million by 2033, growing at a CAGR of 6.4% during the forecast period 2025-2033.
Scleroderma is a rare and intricate autoimmune condition characterized by the abnormal thickening and stiffening of the skin and underlying connective tissues. This disorder is marked by an overactive immune system that triggers excessive production of collagen, leading to the formation of scar tissue (fibrosis) in various parts of the body.
The rising prevalence of scleroderma and advancements in treatment options are driving the market. The scleroderma market is witnessing significant advancements in treatment modalities, particularly with the development of novel drugs, including biologics and targeted therapies. Innovations in the treatment of scleroderma, emphasize their role in improving patient outcomes. These factors have driven the global scleroderma market expansion.
Scleroderma Market Overview
Market Dynamics: Drivers & Restraints
Rising Prevalence of Scleroderma and Advancements in Treatment Options
The demand for the global scleroderma market is driven by multiple factors. One of the primary factors is the rising prevalence of scleroderma and advancements in treatment options. This rise can be attributed to several interconnected elements, including genetic predispositions, environmental factors, and immune system dysfunction.
As per the National Center for Biotechnology Information (NCBI) research publication in March 2023, the global incidence of systemic sclerosis (SSc) is 8.64 cases per 100,000 person-years, while the prevalence is 18.87 cases per 100,000 individuals. This suggests that approximately 670,000 people are newly diagnosed with SSC annually. The publication also says that 83.9% of countries around the world do not have epidemiological data on SSc. The incidence and prevalence rates are higher among females, adults, and in high-income countries.
In addition, government initiatives and key player's product launches & approvals would drive this market growth. For instance, in December 2023, the Scleroderma Research Foundation (SRF), announced that the FDA had cleared its Investigational New Drug (IND) application. This clearance allows SRF to launch the CONQUEST clinical trial platform and begin enrolling patients. Sanofi and Boehringer Ingelheim have agreed to contribute an experimental agent each to the CONQUEST trial, making them the first pharmaceutical partners to commit to this protocol designed to expedite the clinical development of new scleroderma treatments.
The increased awareness among healthcare professionals and also individuals who are prone to the condition is expected to drive market growth. For the treatment of scleroderma, the pharmaceutical industry is constantly creating novel and enhanced treatments. These developments, which include stem cell therapies, gene therapy strategies, and tailored immunomodulatory medications, provide promise for better disease management and can lead to an increase in the number of available therapeutic options. All these factors demand the global scleroderma market. Moreover, the rising demand for novel product development contributes to the global scleroderma market expansion.
Stringent Regulatory Requirements
Stringent regulatory requirements significantly impact the global scleroderma market. The development and approval of new treatments for scleroderma are closely regulated by authorities like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These regulatory processes are often time-consuming and expensive, as they typically require extensive clinical trials to confirm the safety and efficacy of new therapies. As a result, pharmaceutical companies can face delays in bringing potentially life-saving medications to market, affecting the availability of new treatments for patients.
Moreover, varying regulatory standards across countries further complicate the situation. Differences in clinical trial protocols, approval timelines, and reimbursement policies can slow the global introduction of scleroderma treatments. These complexities can pose additional challenges for smaller biotech firms that lack the resources to meet strict regulatory requirements, limiting their ability to innovate and compete. Ultimately, these stringent regulations can hinder the overall growth of the scleroderma market and restrict patient access to effective treatments. Thus, the above factors could be limiting the global scleroderma market's potential growth.
For more details on this report – Request for Sample
Scleroderma Market Segment Analysis
Treatment Type:
The immunosuppressants segment is expected to dominate the global scleroderma market share
The immunosuppressants segment holds a major portion of the global scleroderma market share and is expected to continue to hold a significant portion of the global scleroderma market share during the forecast period. The immunosuppressants segment encompasses a range of drugs that are critical for managing scleroderma. This is particularly important given the autoimmune nature of the disease and its widespread effects on multiple organ systems throughout the body. Immunosuppressants are frequently utilized in the management of systemic sclerosis (SSc), also referred to as scleroderma. While these medications can enhance lung function and skin conditions, they may also lead to significant side effects and necessitate regular laboratory monitoring.
The immunosuppressants segment encompasses a variety of drug classes, including biologics and small-molecule therapies. Biologics these medications are derived from living organisms and specifically target components of the immune system. An instance is Boehringer Ingelheim's Ofev (nintedanib), which has received approval for treating systemic sclerosis with interstitial lung disease.
Traditional immunosuppressants mostly, used drugs in this category include methotrexate and mycophenolate mofetil, which are employed to alleviate symptoms of scleroderma and reduce inflammation. There is a strong pipeline of new immunosuppressant therapies currently in development, aimed at enhancing patient outcomes. Ongoing research is focused on discovering novel drug candidates that can improve the efficacy and safety profiles of existing treatments. This includes investigating new biologics and small molecules that may offer better management options for scleroderma patients.
Immunosuppressants are a critical class of medications used in the treatment of scleroderma, particularly systemic sclerosis. These drugs function by inhibiting or preventing the immune system's activity, which is essential in managing the autoimmune aspects of the disease. Methotrexate (MTX) is a widely used immunosuppressant in the treatment of scleroderma, particularly for managing skin manifestations. Its mechanism of action involves inhibiting DNA synthesis, which has several important implications for reducing the autoimmune activity characteristic of this disease.
Moreover, key players more focus on the treatment for systemic sclerosis and the research studies, rising number of clinical trials that would propel this market growth. As per the University of Alberta in October 2022, researchers have identified genetic mutations in the cells of scleroderma patients that resemble those found in cancer, which may lead to new treatment options for this challenging condition. These findings explain why scleroderma, an incurable skin disease, often does not respond well to traditional immunosuppressive therapies. The study aims to clarify the mechanisms linking abnormal DNA damage responses to the disease's fibrosis and inflammation, potentially paving the way for innovative treatment strategies.
Also, in March 2024, Cabaletta Bio, Inc. announced that the U.S. Food and Drug Administration (FDA) had granted an Orphan Drug Designation (ODD) to CABA-201. This investigational therapy is a fully human CD19-CAR T cell treatment that contains 4-1BB and is being explored for its potential to treat systemic sclerosis (SSc). These factors have solidified the segment's position in the global scleroderma market.
Geographical Analysis
North America is expected to hold a significant position in the global scleroderma market share
North America holds a substantial position in the global scleroderma market and is expected to hold most of the market share. The rising prevalence of systemic sclerosis in North America is a primary driver of market growth. Systemic sclerosis (SSc), also known as scleroderma, is a rare autoimmune disease that affects around 75,000 individuals in the United States. Interstitial lung disease (ILD) is a common complication of SSc, with up to 80% of patients developing this progressive condition. ILD can significantly impair lung function and, in severe cases, may be life-threatening.
In North America, localized scleroderma is becoming more common, much like many other autoimmune illnesses. This is caused by a complicated interaction between environmental and genetic factors. The increasing incidence of scleroderma, particularly localized scleroderma and systemic sclerosis, is driving the demand for effective treatment options globally. Localized scleroderma primarily affects the skin and underlying tissues. It occurs more commonly in children but can also appear in adults. The estimated incidence of localized scleroderma is 2.7 per 100,000 individuals in the U.S.
Systemic sclerosis, also known as systemic scleroderma, affects the skin, tissues, blood vessels, and major organs. The estimated incidence of systemic sclerosis is 1.4 per 100,000 person-years. This means that for every 100,000 people in the population, 1.4 new cases of systemic sclerosis are diagnosed each year. A larger patient base seeking treatment due to more diagnosed cases raises the demand for medications, therapies, and related services. This promotes market expansion in several industries, including healthcare, biotechnology, and pharmaceuticals. The need for early diagnosis means that there will undoubtedly be a growing need for precise and effective diagnostic instruments, such as skin biopsies, blood tests, and imaging methods like MRI and ultrasound.
Moreover, advancements in treatment options, favorable reimbursement policies, key player’s focus on research and development (R&D) activity, and growing healthcare expenditure are driving this market growth in this region. In addition, in this region, a major number of key player's presence, well-advanced healthcare infrastructure, and product launches & approvals would propel this market growth. For instance, in February 2024, Certa Therapeutics announced that the U.S. Food and Drug Administration (FDA) has granted Fast Track Designation to its investigational therapy FT011 for treating systemic sclerosis (scleroderma). This designation comes after the FDA previously granted Orphan Drug Designation to FT011 for the same indication.
Also, in September 2022, The U.S. Food and Drug Administration (FDA) granted Fast Track Designation to efzofitimod, a potential first-in-class immunomodulator, for the treatment of interstitial lung disease associated with systemic sclerosis. Thus, the above factors are consolidating the region's position as a dominant force in the global scleroderma market.
Asia Pacific is growing at the fastest pace in the global scleroderma market share
Asia Pacific holds the fastest pace in the global scleroderma market and is expected to hold most of the market share. The scleroderma market in the Asia-Pacific region is experiencing significant growth due to several factors, including rising prevalence, increasing awareness, improved healthcare infrastructure, and the introduction of innovative therapies. Countries like China, India, Japan, and South Korea are playing leading roles in the development of this market.
With the increasing prevalence of autoimmune diseases, including systemic sclerosis, there is a growing demand for effective treatments in Asia-Pacific including China, Japan, and South Korea. Due to the vast population, the potential patient pool for scleroderma is significant. For instance, according to the study conducted by NIH, the pooled prevalence of SSc-ILD in East Asia was 56%. The SSc-ILD prevalence was higher in China (72%) than in Japan (46%) and Korea (51%).
Despite the advancements, awareness of scleroderma remains relatively low in some parts of the region, leading to delayed diagnosis and treatment. Increasing education and awareness programs could further drive market growth. For instance, on August 24. 2024, National Scleroderma Symposium and Patient Awareness Program, this Symposium intends to provide a unique platform for Rheumatologists and allied professionals from across India to share their experiences update their knowledge, and interact with each other. They anticipate more than 250 participants to join. The program will cover various topics related to Scleroderma and CTD-ILD and will be conducted by renowned experts in the field. This program will be beneficial for the medical community in our Institution and will contribute to the overall improvement of patient care.
International companies are beginning to introduce advanced therapies in this region, although access to biologics remains limited in some areas. New treatments like Ofev (nintedanib) have become available for systemic sclerosis patients in major urban centers. Moreover, generics and biosimilars are helping to improve accessibility at lower costs.
There is an increasing number of clinical trials, including those for autoimmune diseases like scleroderma. Collaborations between pharmaceutical companies and global players with support from regulatory bodies are also bringing innovative treatments to the local market. This further boosts the market growth in the Asia-Pacific region. Thus, the above factors are consolidating the region's position as the fastest-growing force in the global scleroderma market.
Competitive Landscape
The major global players in the scleroderma market include GSK plc, Certa Therapeutics, F. Hoffmann-La Roche Ltd, Boehringer Ingelheim International GmbH., Sun Pharmaceutical Industries Ltd., Novartis AG, Pfizer Inc, Teva Pharmaceuticals USA, Inc., Eli Lilly and Company, and AUROBINDO PHARMA LIMITED among others.
Key Developments
- In February 2023, the U.S. Food and Drug Administration (FDA) granted Orphan Drug Designation (ODD) to belimumab (brand name Benlysta), a B-cell inhibiting monoclonal antibody, for the potential treatment of systemic sclerosis (SSc). Belimumab is a fully human monoclonal antibody that binds to soluble B-lymphocyte stimulator (BLyS), which is found to be elevated in patients with systemic autoimmune diseases like systemic lupus erythematosus (SLE) and lupus nephritis (LN).
Scleroderma Market Scope
| Metrics | Details | |
| CAGR | 6.4% | |
| Market Size Available for Years | 2022-2033 | |
| Estimation Forecast Period | 2025-2033 | |
| Revenue Units | Value (US$ Mn) | |
| Segments Covered | Disease Type | Systemic Sclerosis, Localized Sclerosis |
| Treatment Type | Immunosuppressants, Vasodilators, Proton Pump Inhibitors, Analgesics, Phototherapy, Others | |
| Distribution Channel | Hospitals & Clinics, Retail Pharmacies, Online Pharmacies | |
| Regions Covered | North America, Europe, Asia-Pacific, South America, and Middle East & Africa | |
Why Purchase the Report?
- Pipeline & Innovations: Reviews ongoing clinical trials, and product pipelines, and forecasts upcoming advancements in medical devices and pharmaceuticals.
- Product Performance & Market Positioning: Analyzes product performance, market positioning, and growth potential to optimize strategies.
- Real-World Evidence: Integrates patient feedback and data into product development for improved outcomes.
- Physician Preferences & Health System Impact: Examines healthcare provider behaviors and the impact of health system mergers on adoption strategies.
- Market Updates & Industry Changes: Covers recent regulatory changes, new policies, and emerging technologies.
- Competitive Strategies: Analyzes competitor strategies, market share, and emerging players.
- Pricing & Market Access: Reviews pricing models, reimbursement trends, and market access strategies.
- Market Entry & Expansion: Identifies optimal strategies for entering new markets and partnerships.
- Regional Growth & Investment: Highlights high-growth regions and investment opportunities.
- Supply Chain Optimization: Assesses supply chain risks and distribution strategies for efficient product delivery.
- Sustainability & Regulatory Impact: Focuses on eco-friendly practices and evolving regulations in healthcare.
- Post-market Surveillance: Uses post-market data to enhance product safety and access.
- Pharmacoeconomics & Value-Based Pricing: Analyzes the shift to value-based pricing and data-driven decision-making in R&D.
The global scleroderma market report delivers a detailed analysis with 60+ key tables, more than 50 visually impactful figures, and 176 pages of expert insights, providing a complete view of the market landscape.
Target Audience 2025
- Manufacturers: Pharmaceutical, Medical Device, Biotech Companies, Contract Manufacturers, Distributors, Hospitals.
- Regulatory & Policy: Compliance Officers, Government, Health Economists, Market Access Specialists.
- Technology & Innovation: AI/Robotics Providers, R&D Professionals, Clinical Trial Managers, Pharmacovigilance Experts.
- Investors: Healthcare Investors, Venture Fund Investors, Pharma Marketing & Sales.
- Consulting & Advisory: Healthcare Consultants, Industry Associations, Analysts.
- Supply Chain: Distribution and Supply Chain Managers.
- Consumers & Advocacy: Patients, Advocacy Groups, Insurance Companies.
- Academic & Research: Academic Institutions.
Suggestions for Related Report
- Global Systemic Sclerosis Market
- Global Scleroderma Therapeutics Market
- Global Localized Scleroderma Market
For more pharmaceuticals-related reports, please click here