Liquid Biopsy Early Cancer Detection Market Overview
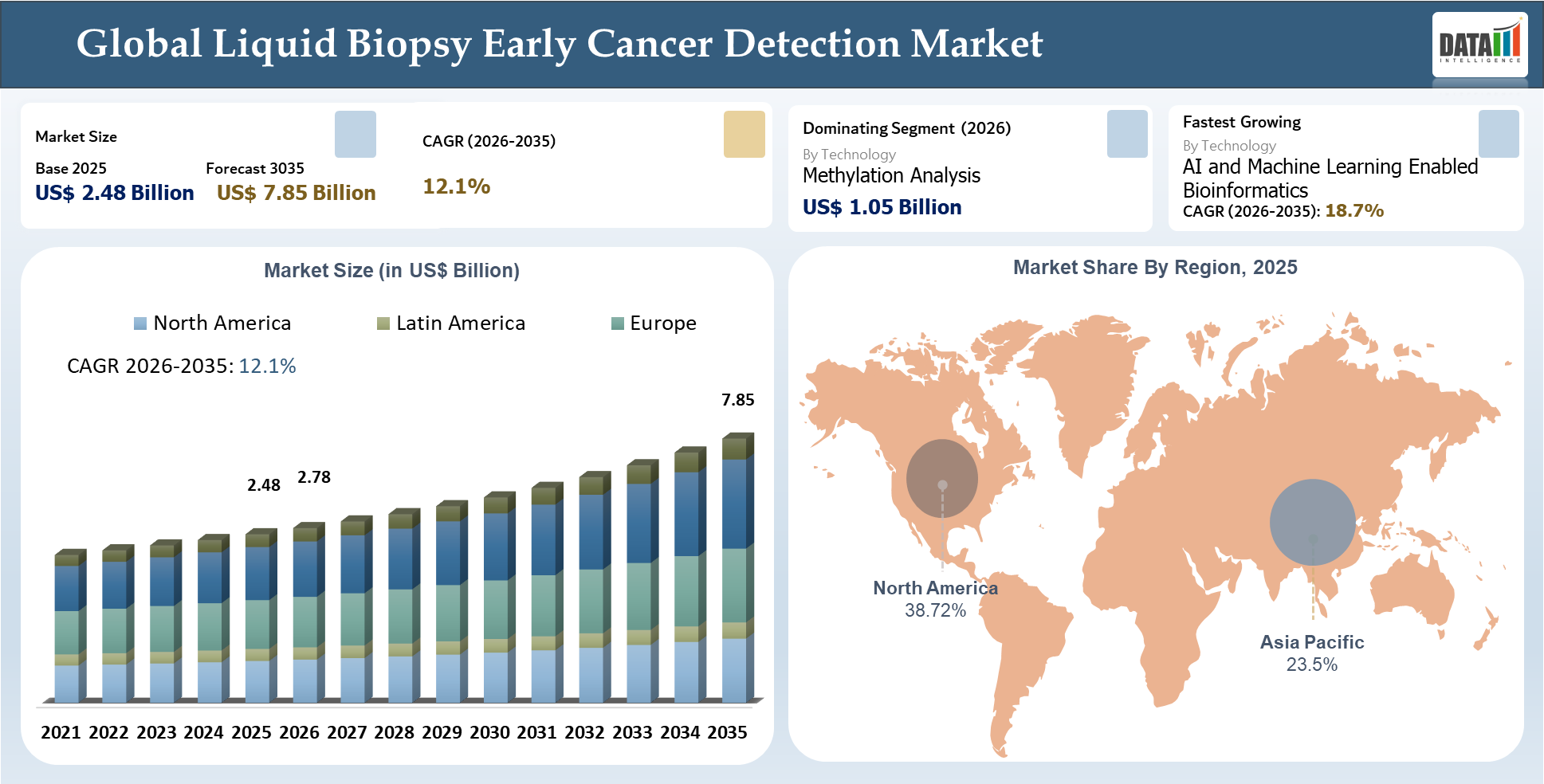
The global liquid biopsy early cancer detection market size reached US$ 2.48 billion in 2025 and is expected to reach US$ 7.85 billion by 2035, growing at a CAGR of 12.1% during the forecast period 2026–2035. The liquid biopsy early cancer detection market growth is being supported by the rising shift toward non-invasive screening technologies that can identify cancer signals at an earlier stage with better patient compliance than conventional diagnostic methods. The market is evolving from single-biomarker approaches toward advanced platforms integrating methylation analysis, multi-omics, next generation sequencing, and AI-enabled bioinformatics, which are improving assay accuracy and tumor-origin prediction.
The liquid biopsy early cancer detection market trends indicate growing interest in multi-cancer early detection tests, broader adoption of blood-based screening models, and stronger focus on preventive healthcare and high-risk population screening. At the same time, the liquid biopsy early cancer detection market challenges opportunity landscape is shaped by reimbursement limitations, false-positive concerns, and the need for stronger clinical workflow integration. Even so, continued innovation in liquid biopsy, increasing demand for early cancer detection, and expansion of scalable cancer detection platforms are expected to strengthen long-term market potential.
Liquid Biopsy Early Cancer Detection Industry Trends and Strategic Insights
- In order to increase the sensitivity of detection in asymptomatic cases and early stages of cancer, there is a high tendency towards methylation and multi-omics based screening platforms in the liquid biopsy early cancer detection market space. Major companies like GRAIL, Guardant Health, and Exact Sciences Corporation are investing heavily in their platforms using blood based multi-cancer and single-cancer detection screening techniques, which increases scalability and relevance.
- For instance, organizations like Freenome Holdings, Inc., DELFI Diagnostics, and Harbinger Health are developing platforms with multi-tiered biomarker detection that includes fragmentomics, proteomics, and computational techniques. This makes performance and platform clinical relevance more valuable compared to the use of a single biomarker.
- Moreover, organizations like SeekIn Inc., Singlera Genomics Inc., Elypta, and AnchorDx are pushing innovation in terms of cost-effective screening techniques, metabolism based detection, and regional-based commercialization strategy. However, clinical validation, reimbursement, physicians' acceptance, and prevention of screening will be key factors deciding the leaders in the growth phase of the liquid biopsy early cancer detection market
Market Scope
| Metrics | Details | |||
| 2025 Market Size | US$ 2.48 Billion | |||
| 2035 Projected Market Size | US$ 7.85 Billion | |||
| CAGR (2026-2035) | 12.1% | |||
| Largest Market | North America | |||
| Fastest Growing Market | Asia-Pacific | |||
| By Test Type | Multi Cancer Early Detection Tests, Single Cancer Early Detection Tests, High Risk Screening Liquid Biopsy Tests | |||
| By Biomarker Typeer | Circulating Tumor DNA (ctDNA), Cell Free DNA Methylation Biomarkers, Cell Free RNA (cfRNA), Circulating Tumor Cells (CTCs), Exosomes and Extracellular Vesicles, Protein Biomarkers, Multiomics Biomarker Panels, Others | |||
| By Sample Type | Blood, Plasma, Serum, Urine, Saliva, Others | |||
| By Technology | Next Generation Sequencing (NGS), Polymerase Chain Reaction (PCR), Digital PCR, Methylation Analysis, Immunoassay Based Detection, AI and Machine Learning Enabled Bioinformatics, Others | |||
| By Cancer Type | Lung Cancer, Colorectal Cancer, Breast Cancer, Prostate Cancer, Liver Cancer, Pancreatic Cancer, Ovarian Cancer, Gastric Cancer, Multi Cancer Screening, Others | |||
| By Application | Population Screening, High Risk Patient Screening, Complementary Screening with Conventional Methods, Recurrence Risk and Residual Disease Assessment | |||
| By End-User | Hospitals, Cancer Specialty Centers, Diagnostic Laboratories, Academic and Research Institutes, Preventive Healthcare Programs, Others | |||
| By Region | North America | U.S., Canada, Mexico | ||
| Europe | Germany, UK, France, Spain, Italy, Poland | |||
| Asia-Pacific | China, India, Japan, Australia, South Korea, Indonesia, Malaysia | |||
| Latin America | Brazil, Argentina | |||
| Middle East and Africa | UAE, Saudi Arabia, South Africa, Israel, Turkiye | |||
| Report Insights Covered | Competitive Landscape Analysis, Company Profile Analysis, Market Size, Share, Growth | |||
Disruption Analysis
Shift from Mutation-Based Testing to Multi-Omics and Methylation-Driven Platforms Reshaping Early Cancer Detection
A disruptive change is happening in the market for the early diagnosis of cancer using the liquid biopsy test because of the shift from the traditional mutation-based diagnostic tools to the methylation-led, and multi-omics-based platforms. Traditional methods were predominantly focused on mutations and could be faced with challenges when trying to detect low tumor fractions in asymptomatic patients. On the other hand, modern methods use DNA methylation, fragmentomics, proteomics, and RNA to achieve better results.
This innovation process is creating new dynamics within the industry as companies that used to depend on single biomarker tests are forced to move to the new detection system. The main emphasis of modern technologies is put on predicting tumor origin, minimizing false-positive results and making population-level screenings possible. The transformation is disruptive for the industry players who cannot afford advanced analysis techniques and AI.
While this is happening, AI-driven bioinformatics software solutions are boosting data analysis capabilities, increasing the precision of diagnoses, and facilitating the scaling of tests. The fusion of multi-omics techniques and analytics is creating new opportunities for businesses to thrive on preventive medicine and regular screenings. Companies unable to follow the emerging trends in the liquid biopsy early cancer detection market will lose out to more innovative organizations entering this competitive market space..
BCG Matrix: Company Evaluation
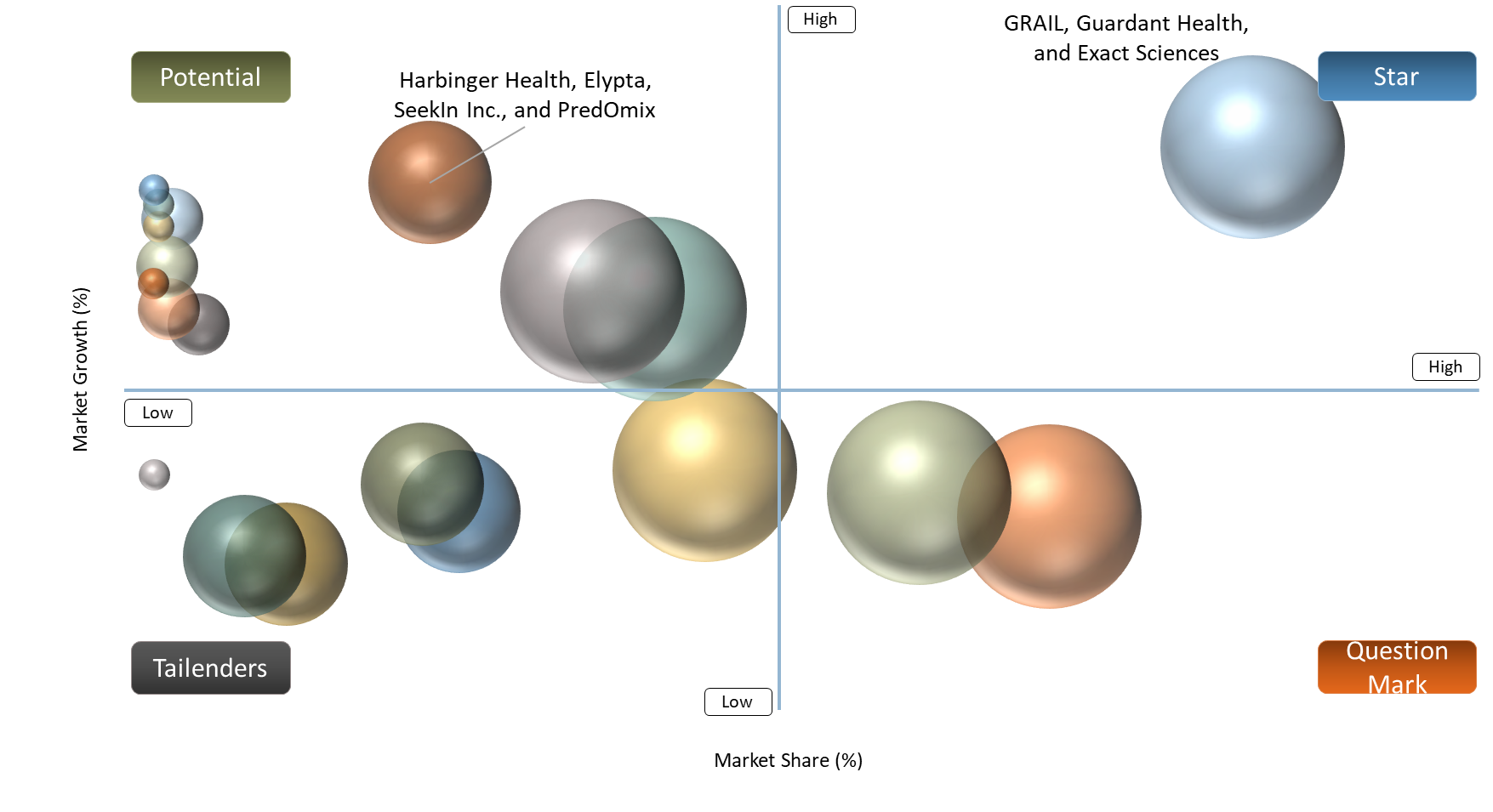
GRAIL, Guardant Health, and Exact Sciences Corporation will be categorized as Stars. The reason behind this is their strong commercial presence and adoption rate in the liquid biopsy early cancer detection market. Additionally, they already have an established presence in the industry due to advanced multi-cancer early detection platforms and physician awareness along with integration in broader early cancer detection pathways.
Harbinger Health, Elypta, SeekIn Inc., and PredOmix are categorized as Potentials due to their high growth potential in the liquid biopsy early cancer detection market. Although they have a relatively small commercial presence in the market, they can become significant players in the future. They have unique biomarker models and analytics enabled through artificial intelligence (AI).
Freenome Holdings, Inc., Natera, Inc., and DELFI Diagnostics will fall under the category of Question Marks. The reasoning behind this is that these companies have a lot of potential and innovation capabilities within the sector. However, they still need to build upon their relevance and commercial presence in the liquid biopsy early cancer detection market. Currently, the market has a growth phase for these companies.
Market Dynamics
Clinical shift toward methylation-led and multi-omics signal enrichment platforms
There is a definitive move in the liquid biopsy early cancer detection market from the mutation-focused approach towards methylation-driven and multi-omics signal enrichment methods due to the necessity for enhancing detection accuracy and efficiency at an early stage. The use of ctDNA mutation analysis is less effective for detecting the presence of cancer when there are minimal amounts of tumor-related DNA because the method is based on mutations that may not be present. However, methylation markers allow for better detection even in trace amounts of tumor-derived DNA.
Biomarker integration in multiple levels, ranging from methylation, fragmentomics, proteomics, and transcriptomics, is now being used by companies to optimize signal generation and minimize false positives. The integration of these multiple omic technologies, along with AI models, enables better tumor origin prediction, which is an essential step for further clinical decision making. Moreover, there is a growing interest in using methylation-based platforms because of their increased capabilities in predicting cancer origins among less-well-screened types of cancers, such as pancreatic and liver cancer.
The use of multiple-omics approaches along with AI models has helped improve test accuracy and increase its clinical application potential.
Physician concern over false positives and unclear diagnostic resolution pathways
Physician reluctance is still a major challenge for the implementation of liquid biopsy cancer early detection tests because of issues surrounding false positives and the absence of standard procedures for the diagnosis resolution process. Early cancer detection tests may detect the presence of cancer without localizing the exact location of the tumor, causing confusion when it comes to further diagnostic workup. The result could be an additional battery of diagnostic tests including imaging studies, tissue biopsies, and referrals to other specialists, which increases patients' stress levels and medical expenses.
As far as the clinical process is concerned, there is currently no universal protocol for how to handle positive test results, especially when dealing with asymptomatic patients. This lack of guidance lowers physicians' confidence in the use of such tests at a wide scale. In addition, the likelihood of overdiagnosis and excessive treatment causes worry for health care providers regarding potential patient harm and ethical considerations.
Segmentation Analysis
The global liquid biopsy early cancer detection market is segmented based on the test type, biomarker type, sample type, technology, cancer type, application, end-user and region.
Methylation Analysis Emerges as the Leading Technology in Liquid Biopsy Early Cancer Detection
Among the technologies within the market of liquid biopsy-based early cancer detection, Methylation Analysis is expected to become the most dominant technology type owing to the superior capability to detect very faint tumor signals in patients who have no symptoms and at an early stage of their diseases. The methylation-based tests have greater capacity to cover cancer signals and provide information about the tissue of origin than mutation-based tests. This makes methylation analysis the most likely technology type to drive the future liquid biopsy-based early cancer detection market.
NGS continues to dominate in terms of being the underlying technology used by players across the industry due to its capacity for comprehensive biomarker analysis and the ability to integrate various types of signal information. PCR and Digital PCR also serve as critical tools for targeted detection applications; however, their importance declines in broad screening scenarios. AI and Machine Learning Enabled Bioinformatics currently represents the most dynamic segment since companies resort to computational tools to integrate information such as methylation, fragmentomics, and proteomics. This trend is driving the development of trends within the field of liquid biopsy-based early cancer detection platforms.
Geographical Penetration
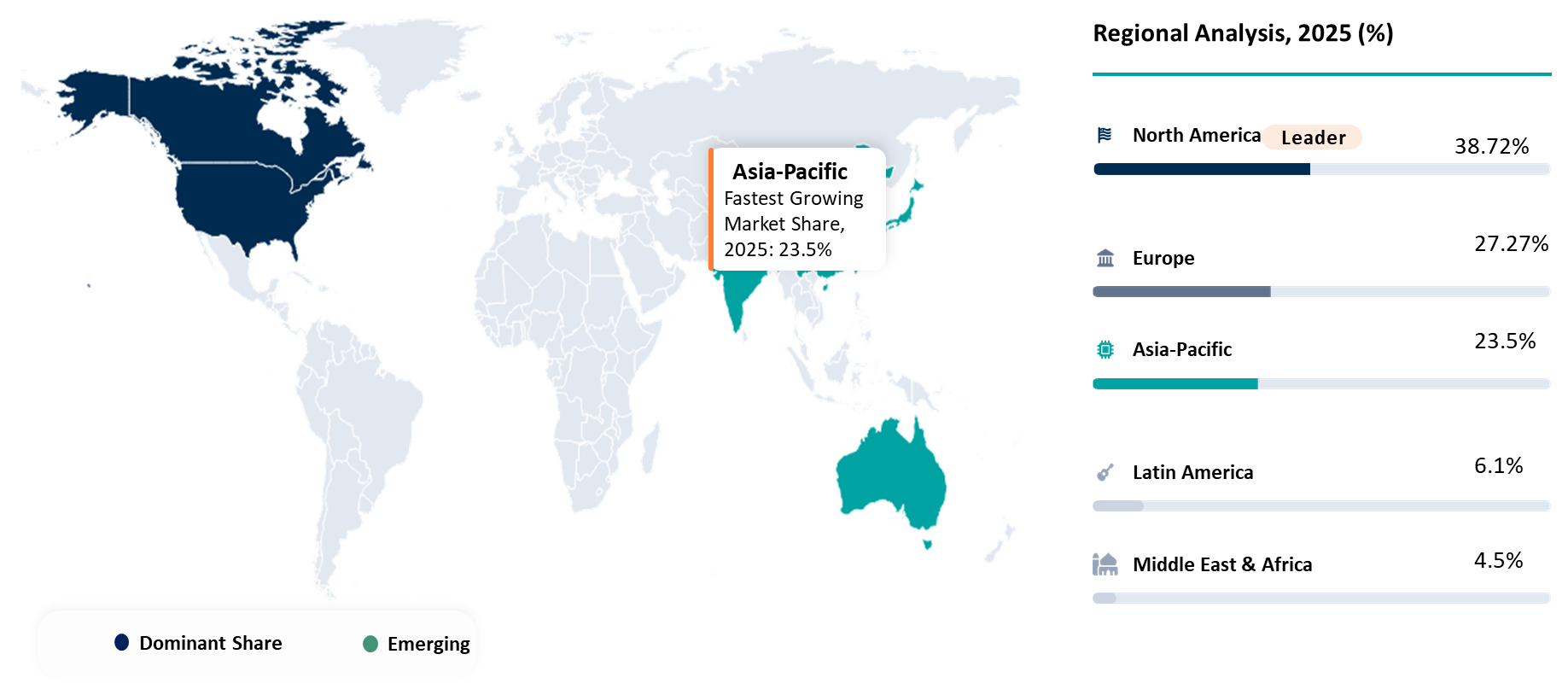
North America Leading Liquid Biopsy Early Cancer Detection Through Early Commercialization and Advanced Diagnostics Infrastructure
North America is currently the leader in terms of liquid biopsy market size for cancer detection, contributing 38.72% of the global revenue generated in 2025 and thereby dominating the geographical segment in the world market. This leadership can be attributed to early commercial launch of blood tests for cancer screening, presence of sound molecular diagnostic technologies, and access to quality oncology treatment facilities in the region. In 2025, the United States accounted for the maximum share of North American liquid biopsy market revenue.
One of the key driving forces in the success of the North American liquid biopsy early cancer detection market is the capability of this region to utilize next generation sequencing, digital PCR processes, and bioinformatics innovations into their oncology and screening processes. Some of the other key factors that have helped this market include an increased need for noninvasive tests, higher incidences of cancers, and the adoption of multi-marker screening programs for cancer detection.
Moreover, North America too enjoys robust evidence generation, innovative environment, and regulation in the United States. Standardization of tests, reimbursement policies, and the challenge of interpreting weak signals are some areas of concern, despite this. Nonetheless, North America continues to hold on to its position as the dominant market player in the field of early detection using liquid biopsy.
U.S. Liquid Biopsy Early Cancer Detection Market Trends
Trends in the U.S. liquid biopsy early cancer detection market continue to be determined by the move from pilot launch to evidence-based scaling up. The increased popularity of multiple cancer early detection tests is one trend. GRAIL applied for FDA pre-market approval for its Galleri test at the beginning of 2026. Exact Sciences reported that in February 2026, Medicare coverage was enabled through federal legislation for multiple cancer early detection tests.
The other significant development within the use of liquid biopsies for early cancer diagnosis is the increased emphasis on incorporating clinical workflow into the process, not just assay innovation. According to the American Cancer Society, MCD tests may identify DNA, RNA, or proteins and even help determine the source of cancer in some cases, but additional processes are required. It is for this reason that specific facilities, like Dana-Farber’s Multi-Cancer Early Detection Clinic, are being developed to assist in the process of making a definitive diagnosis following a positive test.
On the whole, the U.S. market is moving from early adoption to structured commercialization, becoming the model market for deploying early cancer detection systems.
Canada Liquid Biopsy Early Cancer Detection Market Outlook
Canada represents an emerging but strategically important market for liquid biopsy early cancer detection, supported by rising national emphasis on earlier diagnosis, translational cancer research, and broader interest in non-invasive testing pathways. The Canadian Cancer Society has emphasized the need for further liquid biopsy research in Canada in recent years, as it will allow for faster, non-invasive and safer cancer analysis when utilizing blood-, saliva-, and urine-based diagnostic approaches. This development trend will help drive future growth opportunities in the early cancer detection market with regard to liquid biopsy in the country.
The emergence of increasing commercial availability of blood-based cancer tests is one of the major growth drivers for the market forecast. As reported in January 2026, a Canadian release of a blood test used for detecting tumor DNA was made available in Western Canada - indicating early signs of momentum towards commercializing the sector. On top of that, cancer screening strategies remain active in Canada, as reflected by such initiatives as the 2026-2035 Pan-Canadian Lung Cancer Action Plan aimed at timely detection and integration of research findings.
In summary, while not being the largest liquid biopsy early cancer detection market size contributor in North America, Canada remains a promising market on its way to growth.
Competitive Landscape
- The liquid biopsy early cancer detection market is driven primarily by innovation and comprises several types of stakeholders including multi-cancer early detection firms, blood-based cancer screening firms, and biomarker platforms providers. The leading players in this market include GRAIL, Guardant Health, Exact Sciences Corporation, and Freenome Holdings, Inc. who are driving the development of the market in terms of commercialization and implementation of blood-based early cancer detection. The competition in this market is characterized by advancements in methylation testing, next generation sequencing, multi-omics approach, and bioinformatics via artificial intelligence technologies.
- Other significant firms, including SeekIn Inc., Singlera Genomics Inc., Harbinger Health, Elypta, AnchorDx, Lucence Health Inc., Burning Rock Biotech Limited, Natera, Inc., Oxford BioDynamics Plc, PredOmix, Genecast Group Inc., CORE Diagnostics, DELFI Diagnostics, ClearNote Health, ANPAC Bio-Medical Science Co., Ltd., and 20/20 BioLabs, Inc., are intensifying competition in the market through their unique biomarker technologies, regional commercialization strategies, and innovations in platforms. Trends in the liquid biopsy early cancer detection market show that competition is becoming ecosystem-driven, requiring a successful combination of assay performance, biomarker breadth, clinical validation, physician adoption, and scalability of incorporation into standard cancer detection processes.
Key Developments
- January 2026 - GRAIL submitted a Premarket Approval application to the U.S. FDA for Galleri. This is one of the most important regulatory milestones in the liquid biopsy early cancer detection market, as it moves a leading multi-cancer early detection blood test closer to formal FDA review.
- March 2026 - Guardant Health launched Shield MCD in multiple Asian markets through a partnership with Manulife. The launch expanded Guardant’s multi-cancer detection footprint into Hong Kong, Singapore, and the Philippines, reinforcing Asia’s role in liquid biopsy early cancer detection market growth.
- March 2026 - Guardant Health made Shield accessible through Quest Diagnostics’ national ordering and collection network in the U.S. This widened physician access to its FDA-approved blood-based colorectal cancer screening test and strengthened commercialization infrastructure.
- April 2026 - GRAIL announced integration of the Galleri test into the Epic electronic health record platform. This improves ordering workflow and could support broader clinical adoption of liquid biopsy early cancer detection in routine care settings.
- September 2025 - Exact Sciences launched Cancerguard as a commercially available multi-cancer early detection blood test. The company positioned it as the first commercially available MCED test analyzing multiple biomarker classes, making it a notable commercialization milestone in the liquid biopsy space.
- November 2025 - Freenome and Roche expanded their collaboration to develop and commercialize cancer screening tests outside the U.S. The agreement broadened Freenome’s ex-U.S. pathway through a decentralized “kitted” test model.
- October 2025 - SeekIn reported large-scale multi-center validation of OncoSeek and, in January 2026, launched OncoSeek in Costa Rica. Together, these developments show both clinical validation and international commercialization progress for a cost-oriented MCED platform.
- May 2025 - Harbinger Health presented ASCO 2025 data showing clinically meaningful MCED performance in high-risk populations. The company highlighted positive predictive value and early-stage sensitivity for multiple high-mortality cancers, strengthening evidence for its reflex blood-based MCED strategy.
- May 2025 - Elypta became a main partner in a pan-European hereditary multi-cancer screening project. This expanded the role of Elypta’s metabolism-based liquid biopsy platform in hereditary and high-risk cancer screening research across Europe.
Why Choose DataM?
- Technological Innovations and Biomarker Discovery: Highlights cutting-edge innovations in the field of liquid biopsy technology, such as liquid biopsies for the early detection of cancers based on methylation analysis, multi-omics approaches, next-generation sequencing techniques, digital PCR methods, fragmentomics, proteomics, and AI-powered bioinformatics, which have significantly improved the sensitivity, specificity, and ability to predict the origin of tumors in the early diagnosis of cancer.
- Technology Performance and Market Positioning: Analyzes the performance of various competing companies and technologies in multi-cancer early detection and single cancer-screening tests. This assessment is based on parameters such as the sensitivity and specificity of the test, its stage-based efficacy, the capability to predict the tissue of origin, the ease of the procedure, and other important factors.
- Real-World Applications: Provides examples of real-world applications of liquid biopsy in asymptomatic cancer screening, high-risk population screening, cancer prevention initiatives, and complementing existing screening methods.
- Market Updates and Industry Developments: Covers important trends like product launches, FDA filing, validation studies, reimbursement pathway development, partnership announcements, regional launch, and changes in healthcare adoption in North America, Europe, Asia-Pacific, China, and the US.
- Competitor Strategies: Details strategies adopted by key players to establish themselves through innovation in biomarkers, building clinical evidence, engaging physicians, partnering with laboratories and hospitals, launching in multiple regions, and leveraging AI-enabled platforms.
- Pricing Strategy and Market Access: Provides information on pricing strategies used by different organizations for their multi-cancer early detection products and blood-based cancer screening tests. Also covers information on reimbursement, pay-for-private insurance screening, employer-based testing, laboratory access, and regional distribution strategies impacting adoption.
- Market Entry and Growth Opportunities: Highlights new healthcare landscapes to enter into, private diagnostics markets, high-risk screening markets, and preventive care models. In addition, describes the scaling strategies for entry into the liquid biopsy market for early cancer detection based on clinical validation, regulatory pathway, and commercialization strategy.
Target Audience 2026
- Hospitals and Cancer Care Centers: Hospitals specialized in oncology along with specialized clinics utilizing liquid biopsy technology for early cancer detection during screening, diagnosis, and treatment for several types of cancer.
- Diagnostic Laboratories and Pathology Networks: Central and molecular laboratories using the liquid biopsy technology for testing purposes, biomarkers analysis, and screening services for several types of cancer.
- Biotechnology and Diagnostics Companies: Companies involved in liquid biopsy early cancer detection market through developing new platforms such as multi-omics, methylation analysis, and AI-based cancer detection platforms.
- Pharmaceutical and Life Science Companies: Companies that use the early cancer detection tools for their clinical trials and biomarker discovery efforts in precision medicine.
- Investors and Venture Capitalists: Groups who monitor innovations and progress in early cancer detection and growth in multi-cancer early detection platforms.
























































