Market Size
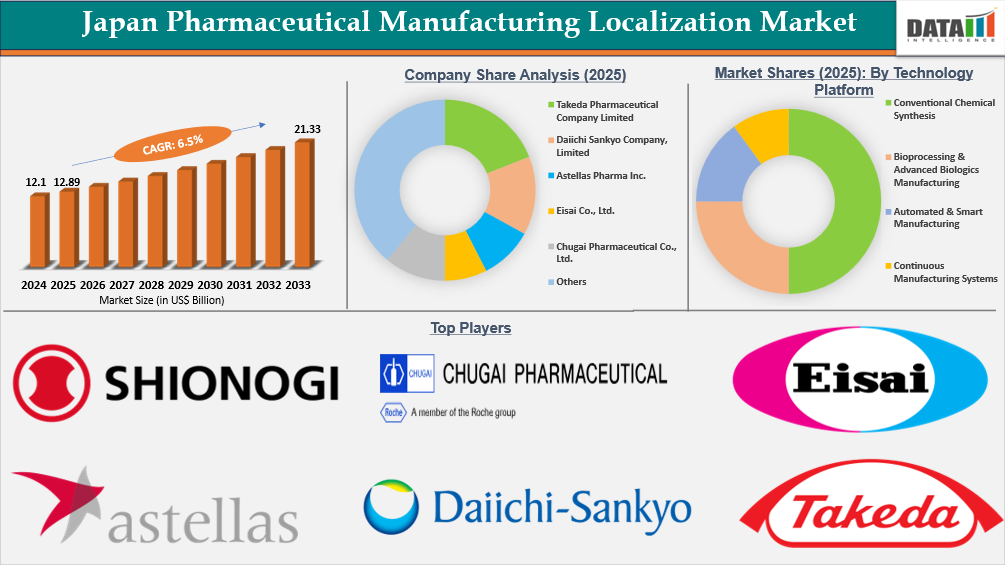
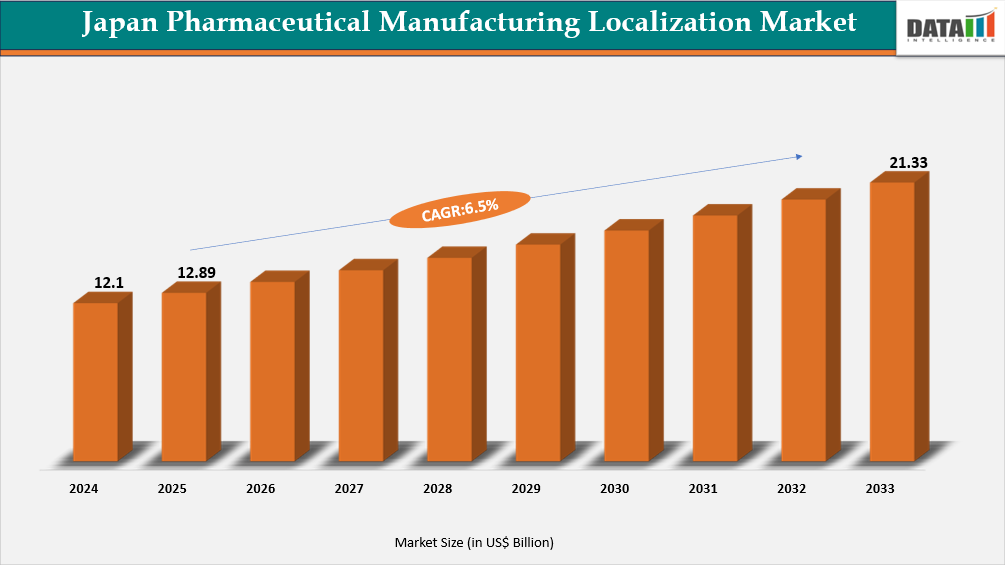
The Japan Pharmaceutical Manufacturing Localization Market reached US$ 12.89 billion in 2025 and is expected to reach US$ 21.33 billion by 2033, growing at a CAGR of 6.5% during the forecast period 2026-2033.
Japan's Pharmaceutical manufacturing Localization Market is gradually developing, owing to strong hospital demand, extensive generic adoption, and high injectable therapy utilization in in-house domestic production and critical care settings. As of 2025, generic medications account for approximately 89% of prescription volume in Japan, reflecting continued government initiatives to control healthcare costs and encourage the substitution of off-patent prescriptions. Hospitals continue to be the principal purchasers of sterile injectables, particularly in inpatient and in-house production settings, resulting in consistent demand for locally manufactured generics to limit treatment costs.
Demographic and medical conditions are also significant drivers. With approximately 29% of the population aged 65 or older, Japan has the greatest rate of cancer, chronic disorders, and age-related diseases, all of which rely largely on injectable therapy. Over one million new cancer cases are recorded each year, and in-house domestic manufacturing continues to dominate the production of sterile injectable generics, particularly chemotherapeutic drugs manufactured in domestic API facilities. These structural healthcare considerations, together with stable reimbursement systems and strong domestic production capabilities, provide a favorable environment for long-term growth in Japan's localized pharmaceutical manufacturing sector.
Industry Trends and Strategic Insights
- By Technology Platform segment, Conventional Chemical Synthesis led the Japan Pharmaceutical Manufacturing Localization Market, capturing the largest revenue share of 50% in 2025.
Japan Pharmaceutical Manufacturing Localization Market Size and Future Outlook
- 2025 Market Size: US$ 12.89 billion
- 2033 Projected Market Size: US$ 21.33 billion
- CAGR (2026–2033): 6.5%
Market Dynamics
Adoption of Advanced Manufacturing Technologies as a Key Driver of Japan’s Pharmaceutical Manufacturing Localization
The use of advanced manufacturing technologies is a major driver of Japan's pharmaceutical manufacturing localization. Domestic facilities can manufacture medications more efficiently, precisely, and consistently by combining automation, robotics, artificial intelligence, digitalization, and continuous manufacturing. These technologies alleviate Japan's labor shortages while maintaining rigorous adherence to PMDA laws and international quality standards. They are especially important for producing complicated goods like biologics, sterile injectables, and cell or gene therapies, which require highly controlled and adaptable manufacturing settings. Furthermore, sophisticated technologies offer scalability, on-demand production, and resource optimization, which reduces errors and operational expenses. As a result, pharmaceutical corporations are boosting their investments in high-tech local facilities, bolstering Japan's status as a hub for complex and dependable medication manufacture.
Segmentation Analysis
The Japan Pharmaceutical Manufacturing Localization Market is segmented based on product category, manufacturing model, technology platform, therapeutic focus, facility type & capacity and end user.
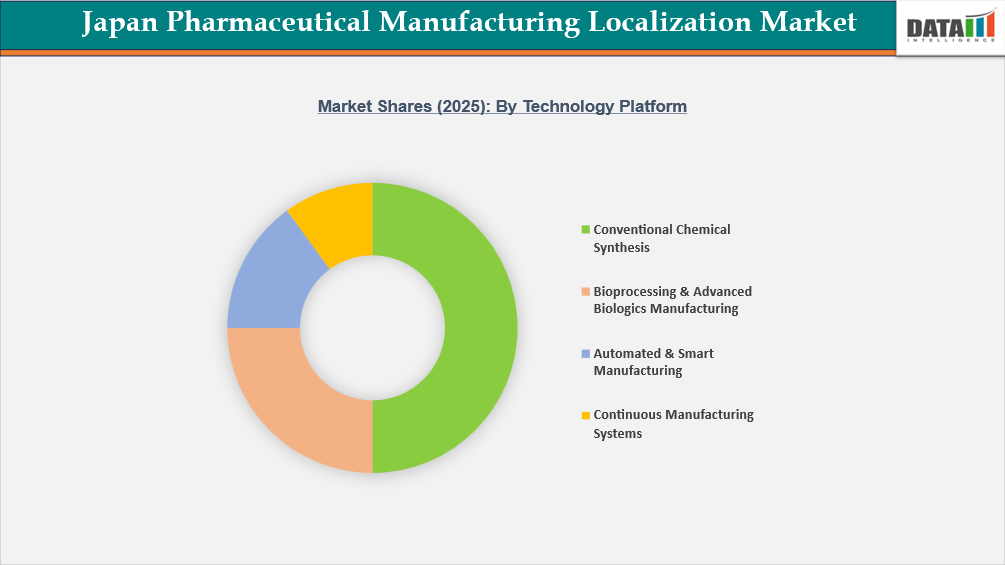
Conventional Chemical Synthesis in Technology Platform Drives Sustained Growth for Japan Pharmaceutical Manufacturing Localization Market
Japan Pharmaceutical Manufacturing Localization Market is most greatly powered by conventional chemical synthesis, as it primarily supports both Japan's overall volume of pharmaceuticals and its infrastructure. 70-75% of total prescription volume in Japan consists of small-molecule drugs manufactured through chemical synthesis; in addition, nearly 85-90% of generic drugs dispensed in Japan have also been chemically synthesized. As generics represent such a large percentage of pharmaceuticals dispensed by Japanese pharmacies, this creates an even stronger case for the continued prevalence of chemical synthesis as part of the localized pharmaceutical manufacturing environment. An estimated 60-65% of drug manufacturing facilities used to produce pharmaceuticals in Japan have been primarily configured for using batch-mode API synthesis methods while producing oral solid dosage forms, which have required decades' worth of capital investment in reactor-type processes. From a localization standpoint, this platform has strategic value, as essential therapeutic categories such as cardiovascular, CNS health, anti-diabetic, and anti-infective medications comprise >65% of routine chronic prescriptions issued by physicians in Japan while being composed mostly of small-molecule compounds. Additionally, chemical batch manufacturing generally provides a larger quantity of product manufacturing than any other type of medicine, provides the lowest cost per unit for pharmaceutical products, and meets price-sensitive buyers through generic supply; therefore, when reviewing the pharmaceutical manufacturing localization market in Japan, the continued dominance of this platform will be increasingly driven by production volume, installed capacity, therapeutic range, and cost effectiveness of supply.
Competitive Landscape
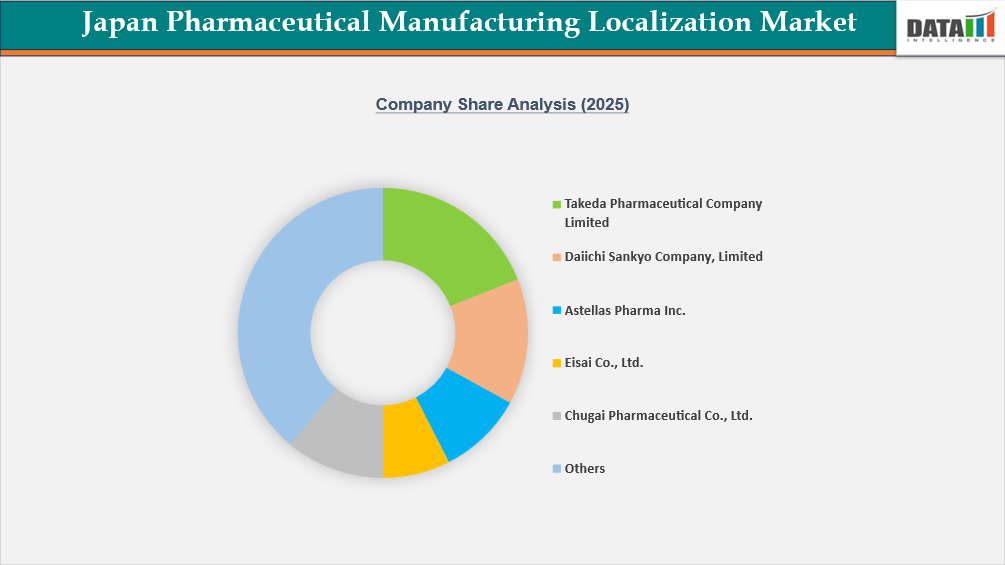
The competitive landscape of the Japan Pharmaceutical Manufacturing Localization Market is moderately consolidated. Mainly domestic pharmaceuticals have the capacity to manufacture products in-house, and these firms account for the majority of all APIs, biologics, or sterile injectables that are manufactured within Japan. Major players such as Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company, Limited, and Astellas Pharma Inc. operate with vertically integrated manufacturing facilities and advanced production capabilities. Other significant manufacturers of pharmaceuticals with a strong domestic presence in their production of specialized therapeutics include Eisai Co., Ltd., Otsuka Pharmaceutical Co., Ltd., and Mitsubishi Tanabe Pharma Corp. Some companies that have less direct competition but help to provide competitively priced biologics or develop a biologic's manufacturing capability are Chugai Pharmaceutical Co., Ltd., CMIC HOLDINGS Co., LTD., Nipro Corporation, and Shionogi & Co., Ltd., in addition to being providers for contract manufacturers of sterile injectables or active raw materials. The competitive position of pharmaceutical manufacturers throughout Japan is strategically based on the continued expansion of production in biologics, sterile injectables, biosimilars, and localized API production facilities.
Key Developments
- In August 2025, Takeda Pharmaceutical Company Limited formally adopted and began operating an AI-based demand forecasting system in Japan's domestic production and supply operations. The solution improves production planning accuracy, optimizes inventory management, and ensures a consistent pharmaceutical supply across the country, supporting Takeda's localization and digital manufacturing strategy.
- In March 2025, Astellas Pharma Inc. inked a binding agreement with Yaskawa Electric Corporation to form a joint venture in Japan centered on automated cell therapy manufacturing. The new company, which will be formally established in September 2025, intends to improve scalability, precision, and efficiency by integrating modern robotic technology with cell cultivation methods. The plan was officially announced in 2025 and is currently in the establishment phase, with full operational activities expected after regulatory procedures are completed.
What Sets This Japan Pharmaceutical Manufacturing Localization Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, covering Japan demand by key segmentation, with detailed analysis.
- Regulatory Intelligence – comprehensive analysis of the regulatory frameworks in Japan that affect local pharmaceutical production, including adherence to the standards set by the Pharmaceuticals and Medical Devices Agency, clinical trial procedures, labeling specifications, patent exclusivity, quality assurance, and post-marketing surveillance that are pertinent to domestic production.
- Competitive Benchmarking – structured assessment of the top domestic and international companies doing business in Japan, taking into account factors such product lines, pipeline capabilities, geographic reach, pricing policies, technological uniqueness, and alliances that promote regional production.
- Actionable Strategies & Cost Dynamics – Supported by viewpoints from industry experts, regulatory advisers, and Japanese pharmaceutical executives, this report offers strategic insights into lifecycle management, generic entry risks, combination therapy positioning, pricing pressures, and domestic manufacturing cost structures.
























































