Alopecia Areata Market Size
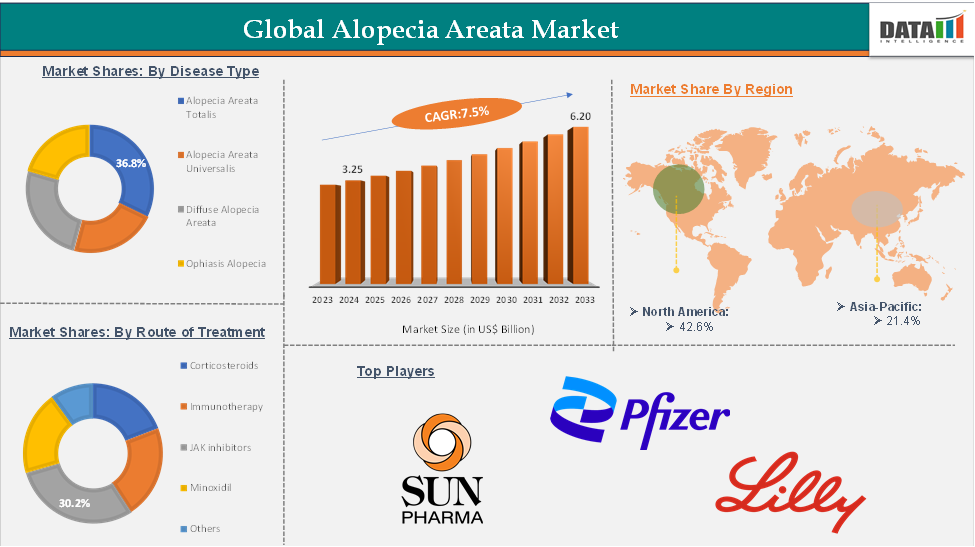
In 2023, the global alopecia areata was valued at US$ 3.02 Billion. The global alopecia areata market size reached US$ 3.25 Billion in 2024 and is expected to reach US$ 6.20 Billion by 2033, growing at a CAGR of 7.5% during the forecast period 2025-2033.
Alopecia Areata Market Overview
Alopecia areata is an autoimmune disorder in which the immune system mistakenly attacks hair follicles, leading to hair loss. The Alopecia Areata market is experiencing significant growth, driven by increasing awareness, rising prevalence, and advancements in immunomodulatory therapies, particularly Janus kinase (JAK) inhibitors. The recent FDA approvals of oral treatments have marked a pivotal shift toward targeted therapies, addressing an unmet need in moderate to severe cases.
Growing research investments, expanding clinical pipelines, and favorable regulatory pathways present strong opportunities for market expansion. Additionally, the rise in autoimmune disorders further fuels demand.
North America dominates the global market due to its advanced healthcare infrastructure, early adoption of novel treatments, and high diagnosis rates. Continued innovation, coupled with strategic partnerships and patient advocacy efforts, is expected to drive the region’s market growth.
Alopecia Areata Market Executive Summary
Alopecia Areata Market Dynamics: Drivers & Restraints
The rising prevalence of alopecia is significantly driving the alopecia areata market growth
The rising prevalence of alopecia areata is a key driver of market growth, as the condition continues to affect a significant portion of the global population. For instance, according to the National Alopecia Areata Foundation, nearly 7 million people in the United States and approximately 160 million individuals worldwide have had, currently have, or will develop alopecia areata. Around 700,000 people in the U.S. are currently living with some form of the disease.
Additionally, about 2% of people across the world are expected to experience alopecia areata at some point in their lifetime. This growing patient population is increasing demand for effective treatments, fueling research, regulatory activity, and commercial interest in innovative therapies.
Increasing innovation in medication is significantly driving the alopecia areata market growth
Increasing innovation in alopecia areata is significantly transforming the treatment landscape and supporting the market’s growth. While traditional therapies like minoxidil and finasteride have been mainstays for years, recent advancements have introduced more precise and effective options. For instance, the emergence of Janus kinase (JAK) inhibitors has provided new possibilities for treating alopecia areata, a type of autoimmune hair loss that was previously difficult to manage.
For instance, in June 2022, Eli Lilly and Company and Incyte (NASDAQ: INCY) announced that the U.S. Food and Drug Administration (FDA) had approved OLUMIANT (baricitinib), a once-daily oral tablet, as the first systemic treatment specifically for adults with severe alopecia areata (AA).
These drugs work by targeting specific immune pathways, offering better results for patients with severe or treatment-resistant cases. In parallel, ongoing research into stem cell therapy, gene-based treatments, and advanced drug delivery systems is improving the safety and effectiveness of interventions.
Restraint:
The high cost of newly approved therapies is hampering the growth of the alopecia areata market
The high cost of newly approved therapies poses a significant challenge to the growth of the alopecia areata market. For instance, Olumiant, approved for severe alopecia areata, may not be accessible to many patients due to limited insurance reimbursement or high out-of-pocket costs. The list price of Olumiant is $2,767.39 for a 30-day supply of 2 mg tablets or $5,534.78 for a 30-day supply of 4 mg tablets.
This financial barrier can restrict patient access, reduce treatment uptake, and hinder market expansion, particularly in low- and middle-income regions or among uninsured populations..
Opportunity:
Expansion of clinical trials and novel drug candidates is expected to create a lucrative opportunity for the growth of the alopecia areata market
The expansion of clinical trials and the emergence of novel drug candidates present a major opportunity for the alopecia areata market. As understanding of the disease’s underlying immune mechanisms deepens, more targeted therapies are entering various stages of development. This growing pipeline reflects increased investment and interest from pharmaceutical and biotech companies.
With several first-in-class and next-generation therapies showing promising results, the market is poised for significant growth.
For more details on this report, Request for Sample
Alopecia Areata Market, Segment Analysis
The global alopecia areata market is segmented based on disease type, treatment type, route of administration, and region.
The JAK inhibitors from the treatment type segment are expected to hold 30.2% of the market share in 2024 in the alopecia areata market
JAK inhibitors have taken a leading role in the treatment of alopecia areata, especially in moderate to severe cases. For instance, in July 2024, Sun Pharmaceutical Industries obtained approval from the U.S. Food and Drug Administration (FDA) for LEQSELVI (deuruxolitinib), a medication used to treat adult autoimmune disorders. These treatments have shown strong results in terms of hair regrowth and are generally well-tolerated, making them increasingly popular among both doctors and patients.
Similarly, in June 2022, Eli Lilly and Company and Incyte announced that the U.S. Food and Drug Administration (FDA) had approved OLUMIANT (baricitinib), a once-daily pill, as a first-in-disease systemic treatment for adults with severe alopecia areata (AA), available as 4-mg, 2-mg, and 1-mg tablets.
Additionally, the high prevalence of androgenetic alopecia and alopecia areata among both men and women in the U.S. and Canada contributes to sustained demand for effective treatment options. While corticosteroids and immunotherapy are still commonly used, they often come with drawbacks like side effects or limited long-term success.
Alopecia Areata Market, Geographical Analysis
North America is expected to dominate the global alopecia areata market with a 42.6% share in 2024
North America is expected to dominate the Alopecia Areata market due to a combination of strong healthcare infrastructure, high consumer awareness, and significant investment in research and development. The region has a well-established pharmaceutical industry, with key players actively engaged in developing and commercializing advanced hair loss treatments, including innovative therapies like JAK inhibitors.
For instance, in June 2023, Pfizer Inc. announced that the U.S. Food and Drug Administration (FDA) had approved LITFULO (ritlecitinib), a once-daily oral medication, for the treatment of severe alopecia areata in individuals aged 12 and older. The recommended approved dose is 50 mg. Notably, LITFULO is the first and only FDA-approved treatment specifically indicated for adolescents (12+) with severe alopecia areata.
There is a growing focus on the development of advanced solutions in the region, and with the presence of a large number of pharmaceutical and biopharmaceutical companies, new solutions are emerging. For instance, Arcutis Biotherapeutics, Inc. announced the completion of enrollment for its Phase 1b study of ARQ-255.
This investigational treatment is a topical suspension of ivarmacitinib, a potent and selective JAK1 inhibitor, designed for alopecia areata. Formulated using Arcutis’ proprietary 4D technology, ARQ-255 is engineered to deliver the drug deep into the skin, targeting the base of the hair follicle—the primary site of inflammation in alopecia areata.
The North America market also benefits from a rising focus on aesthetic health and personal grooming, which drives consumer willingness to spend on hair restoration products and services. Furthermore, strong marketing strategies and the popularity of over-the-counter brands in the region contribute to greater product penetration.
Asia-Pacific is growing at the fastest pace in the alopecia areata market holding 21.4% of the market share
Asia-Pacific is seeing the fastest growth in the alopecia areata market, driven by a mix of rising disease prevalence and evolving lifestyles. With more people experiencing autoimmune conditions like alopecia areata, the demand for effective treatment is rising. Rapid urbanization and better awareness of skin and hair health are prompting more individuals to seek medical help.
At the same time, countries like China, Japan, South Korea, and India are ramping up investments in healthcare systems, dermatology research, and advanced treatment options, creating strong momentum for market expansion across the region.
Alopecia Areata Market Competitive Landscape
Top companies in the alopecia areata market include Pfizer Inc., Sanofi, Merck & Co., Takeda Pharmaceutical Company Limited, Indian Immunological Limited, Bharat Biotech, Panacea Biotec, Sinovac Biotech, AstraZeneca, Novartis AG, among others.
Alopecia Areata Market Key Developments
In April 2025, Q32 Bio Inc. (Nasdaq: QTTB) announced that the FDA granted Fast Track designation to bempikibart (ADX-914) for the treatment of alopecia areata. Bempikibart is a fully human anti-IL-7Rα antibody that blocks IL-7 and TSLP signaling to modulate adaptive immune function. It is currently in Phase 2 clinical development.
In February 2025, Nektar Therapeutics announced the completion of target enrollment for its Phase 2b REZOLVE-AA study evaluating rezpegaldesleukin in patients with severe-to-very-severe alopecia areata.
In February 2025, Mallia Therapeutics announced a partnership with Northway Biotech (NBT), a biologics CDMO, to develop the production process and manufacturing of Mallia’s soluble CD83 protein (sCD83). This therapy is designed to promote hair follicle formation and growth.
In March 2025, Alys Pharmaceuticals, Inc. announced that the first patient had been dosed in its Phase IIa trial of ALY-101 for the treatment of alopecia areata (AA), a chronic autoimmune disorder associated with substantial unmet medical need and significant impact on quality of life.
In September 2024, Arcutis Biotherapeutics, Inc. announced the completion of enrollment for its Phase 1b study of ARQ-255, a topical suspension of ivarmacitinib for the treatment of alopecia areata.
Alopecia Areata Market Scope
Metrics | Details | |
CAGR | 7.5% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Bn) | |
Segments Covered | Disease Type | Alopecia Areata Totalis, Alopecia Areata Universalis, Diffuse Alopecia Areata, Ophiasis Alopecia |
Treatment Type | Corticosteroids, Immunotherapy, JAK inhibitors, Minoxidil, Others | |
Route of Administration | Oral, Topical, Intralesional | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
The global alopecia areata market report delivers a detailed analysis with 60+ key tables, more than 55+ visually impactful figures, and 178 pages of expert insights, providing a complete view of the market landscape.