AI-Powered Medical Devices Market Size and Growth
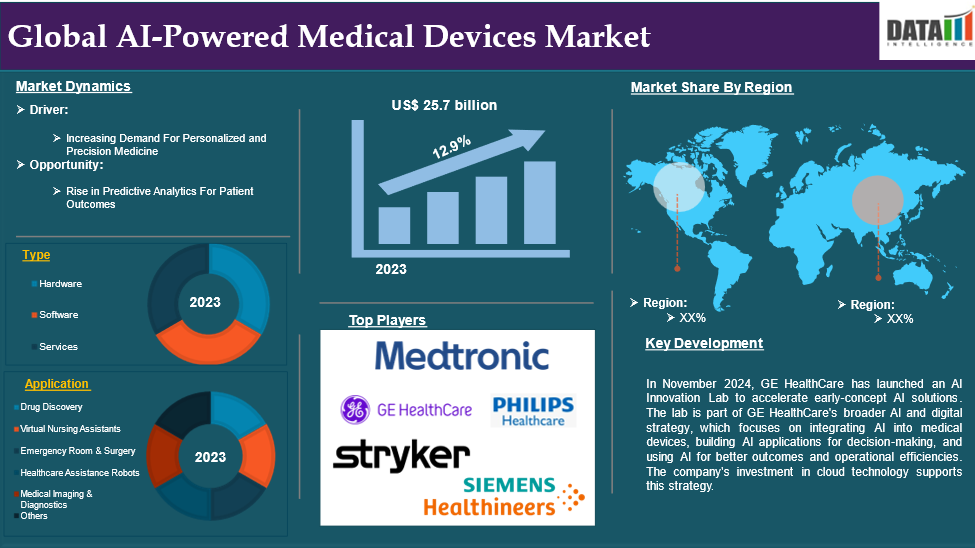
The global AI-powered medical devices market reached US$ 29.0 billion in 2025 and is expected to reach US$ 84.8 billion by 2033, growing at a CAGR of 12.9% during the forecast period 2026-2033.
AI-powered medical devices are revolutionizing healthcare by utilizing AI technologies like machine learning, deep learning, and natural language processing to improve medical diagnosis, treatment, and patient monitoring. These devices analyze vast data, provide real-time insights, and automate complex tasks, enhancing efficiency and patient outcomes. From advanced imaging systems to wearable devices for continuous health monitoring, AI-powered medical devices are reshaping personalized and preventive care, optimizing hospital workflows, and driving a new era in medicine, enabling faster decision-making and more effective treatment strategies.
Executive Summary
AI-Powered Medical Devices Market Dynamics: Drivers & Restraints
Increasing Demand For Personalized and Precision Medicine
The global AI-powered medical devices market is driven by the growing demand for personalized and precision medicine, which is revolutionizing patient care. Advancements in artificial intelligence technologies enable healthcare providers to analyze vast datasets, including genetic information, medical history, lifestyle factors, and real-time health metrics, to deliver highly customized treatment plans. AI algorithms integrated into wearable medical devices can monitor a patient's health parameters continuously, alert clinicians about potential issues before they become critical, and improve diagnostic accuracy in diagnostic imaging. These capabilities not only enhance patient outcomes but also optimize resource allocation by reducing unnecessary tests and hospitalizations.
As chronic diseases and aging populations rise globally, the need for precise and proactive care becomes increasingly critical. Healthcare policies and incentives in regions like North America and Europe encourage the adoption of AI-driven solutions. As the adoption of precision medicine grows, the integration of AI-powered medical devices into routine healthcare practices is expected to accelerate, driving market expansion significantly.
Regulatory and Ethical Challenges Associated with AI Integration in Healthcare
The global AI-powered medical devices market faces regulatory and ethical challenges due to the evolving nature of AI. Regulatory bodies like the FDA and EMA have established rigorous processes for approving advanced AI technologies. However, the dynamic nature of AI makes it difficult to evaluate these devices under static standards, leading to uncertainty and potential delays in product development. Ethical concerns around data privacy and security further complicate the situation, as AI-powered devices rely on vast amounts of patient data, raising concerns about data collection, storage, and usage. The risk of bias in AI algorithms can lead to inaccurate or inequitable outcomes. Collaboration between healthcare providers, technology developers, and policymakers is needed to address these issues, but progress in establishing universally accepted standards has been slow.
AI-Powered Medical Devices Market Segmentation
The global AI-powered medical devices market is segmented based on product type, technology application, end user and region.
Product type:
Hardware segment is expected to dominate the AI-powered medical devices market share
The hardware segment holds a major portion of the AI-powered medical devices market share and is expected to continue to hold a significant portion of the AI-powered medical devices market share during the forecast period.
The global AI-powered medical devices market relies heavily on hardware, including processors, sensors, and accelerators, for high-speed data processing and computational power. GPUs and TPUs are commonly used in AI-powered medical imaging devices for real-time image analysis and pattern recognition. Specialized sensors in wearable medical devices collect vital patient data, which AI systems analyze for actionable insights. Edge computing hardware integrates with these devices, allowing local data processing without cloud computing, ensuring faster responses and improved data security. The development of compact, energy-efficient hardware has made medical devices more portable and accessible for clinical and homecare settings. As AI-powered medical devices become more sophisticated, advancements in hardware technology will continue to drive their performance, scalability, and adoption across diverse healthcare applications.
Application:
Drug Discovery segment is the fastest-growing segment in AI-powered medical devices market share
The drug discovery segment is the fastest-growing segment in the AI-powered medical devices market share and is expected to hold the market share over the forecast period.
Drug discovery is a key driver in the AI-powered medical devices market, as it streamlines the traditional process of drug development. Advanced AI-powered devices can analyze large datasets, including genomic information, chemical compound libraries, and clinical trial results, to identify potential drug candidates more efficiently. These devices help researchers pinpoint molecular targets, predict drug efficacy, and identify side effects early in the development cycle, reducing the time required for new drugs to market. AI-driven systems also enable personalized medicine by integrating patient-specific data, allowing for drug design tailored to individual genetic profiles and disease conditions. This synergy between AI and drug development is increasingly recognized by pharmaceutical companies and healthcare providers, positioning drug discovery as a critical driver in the growth of the AI-powered medical devices market.
AI-Powered Medical Devices Market Geographical Analysis
North America is expected to hold a significant position in the AI-powered medical devices market share
North America holds a substantial position in the AI-powered medical devices market and is expected to hold most of the market share due to its robust healthcare infrastructure, advanced technologies, and supportive regulatory frameworks. Major players like Medtronic, GE Healthcare, and IBM Watson Health are leading the way in integrating AI into medical devices. The region's high adoption rate, research funding, and government initiatives like FDA's streamlined approval processes accelerate market growth. The increasing prevalence of chronic diseases and geriatric population also drive demand for AI-driven diagnostic and monitoring solutions.
Europe is growing at the fastest pace in the AI-powered medical devices market
Europe holds the fastest pace in the AI-powered medical devices market and is expected to hold most of the market share due to its focus on innovation, research collaborations, and high healthcare quality standards. Countries like Germany, the UK, and France have invested heavily in AI research, particularly in medical imaging and robotics. The European Union's Horizon Europe program supports AI adoption through funding and partnerships. The aging population and increasing healthcare burden in Europe also drive demand for efficient diagnostic and therapeutic solutions. Regulatory advancements like the EU Medical Device Regulation promote innovation in AI-based solutions. Both public and private healthcare systems are incorporating AI to reduce costs and improve efficiency.
Competitive Landscape
The major global players in the AI-powered medical devices market includeStryker Corporation, Medtronic, GE Healthcare, Philips Healthcare, Siemens Healthineers, Johnson & Johnson, Micron Technology Inc, Nvidia Corporation, Caption Health, Augmedix, Inc among others.
Recent Developments
- March 2026 – Philips Healthcare deploys AI-driven imaging platform
Philips expanded its AI-powered radiology and cardiac imaging platform to hospitals in Europe and North America, improving diagnostic accuracy and workflow efficiency. - March 2026 – GE Healthcare integrates AI into ultrasound devices
GE Healthcare launched next-generation AI algorithms for ultrasound systems that assist clinicians in automated measurements, anomaly detection, and enhanced imaging guidance for cardiac and fetal exams. - February 2026 – Siemens Healthineers expands AI-driven lab diagnostics solutions
Siemens introduced AI-based algorithms for lab diagnostics and imaging analytics, enabling faster detection of critical biomarkers and more efficient clinical decision support in hospitals. - February 2026 – Stryker Corporation advances surgical navigation with AI analytics
Stryker integrated AI-assisted analytics into its surgical navigation systems, improving intraoperative guidance and predictive outcomes for orthopedic and neurosurgical procedures. - January 2026 – Medtronic enhances AI for cardiac rhythm management
Medtronic deployed AI-powered remote monitoring and predictive analytics tools in cardiac devices to detect arrhythmias and optimize patient therapy, improving early intervention outcomes. - January 2026 – Caption Health receives FDA clearance for AI ultrasound guidance
Caption Health’s AI platform for echocardiography received expanded FDA clearance for automated image acquisition and interpretation, enabling less experienced clinicians to perform high-quality cardiac exams.
Emerging Players
The emerging players in the AI-powered medical devices market include Zebra Medical Vision, Aidoc, Butterfly Network, HeartFlow, Viz.ai and among others.
Report Insights
| Metrics | Details | |
| CAGR | 12.9% | |
| Market Size Available for Years | 2025-2033 | |
| Estimation Forecast Period | 2026-2033 | |
| Revenue Units | Value (US$ Bn) | |
| Volume (Units) | ||
| Segments Covered | Product type | Hardware, Software, Services |
| Technology | Machine Learning, Deep Learning, Natural Language Processing (NLP), Computer Vision, Predictive Analytics | |
| Application | Drug Discovery, Virtual Nursing Assistants, Emergency Room & Surgery, Healthcare Assistance Robots, Medical Imaging & Diagnostics, Others | |
| End User | Hospitals and Clinics, Diagnostic Centers, Research Institutes, Ambulatory Surgical Centers (ASCs), Homecare Settings | |
| Regions Covered | North America, Europe, Asia-Pacific, South America, and Middle East & Africa | |
Why Purchase the Report?
- Pipeline & Innovations: Reviews ongoing clinical trials, product pipelines, and forecasts upcoming advancements in medical devices and pharmaceuticals.
- Product Performance & Market Positioning: Analyzes product performance, market positioning, and growth potential to optimize strategies.
- Real-World Evidence: Integrates patient feedback and data into product development for improved outcomes.
- Physician Preferences & Health System Impact: Examines healthcare provider behaviors and the impact of health system mergers on adoption strategies.
- Market Updates & Industry Changes: Covers recent regulatory changes, new policies, and emerging technologies.
- Competitive Strategies: Analyzes competitor strategies, market share, and emerging players.
- Pricing & Market Access: Reviews pricing models, reimbursement trends, and market access strategies.
- Market Entry & Expansion: Identifies optimal strategies for entering new markets and partnerships.
- Regional Growth & Investment: Highlights high-growth regions and investment opportunities.
- Supply Chain Optimization: Assesses supply chain risks and distribution strategies for efficient product delivery.
- Sustainability & Regulatory Impact: Focuses on eco-friendly practices and evolving regulations in healthcare.
- Post-market Surveillance: Uses post-market data to enhance product safety and access.
- Pharmacoeconomics & Value-Based Pricing: Analyzes the shift to value-based pricing and data-driven decision-making in R&D.
The global AI-powered medical devices market report delivers a detailed analysis with 60+ key tables, more than 50 visually impactful figures, and 176 pages of expert insights, providing a complete view of the market landscape.
Target Audience 2026
- Manufacturers: Pharmaceutical, Medical Device, Biotech Companies, Contract Manufacturers, Distributors, Hospitals.
- Regulatory & Policy: Compliance Officers, Government, Health Economists, Market Access Specialists.
- Component & Innovation: AI/Robotics Providers, R&D Professionals, Clinical Trial Managers, Pharmacovigilance Experts.
- Investors: Healthcare Investors, Venture Fund Investors, Pharma Marketing & Sales.
- Consulting & Advisory: Healthcare Consultants, Industry Associations, Analysts.
- Supply Chain: Distribution and Supply Chain Managers.
- Consumers & Advocacy: Patients, Advocacy Groups, Insurance Companies.
- Academic & Research: Academic Institutions.