Global Achondroplasia Market: Industry Outlook
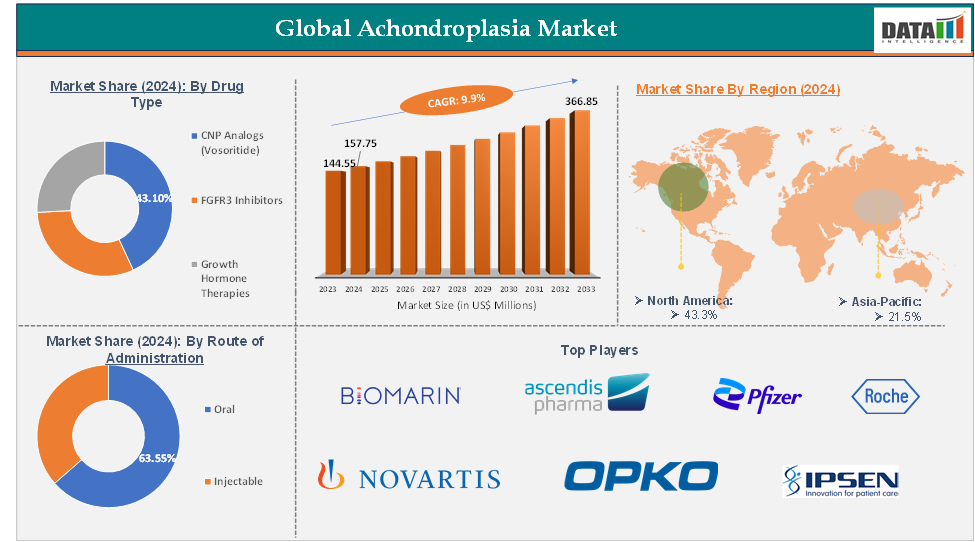
The global achondroplasia market reached US$ 144.55 Million in 2023, with a rise of US$ 157.75 Million in 2024 and is expected to reach US$ 366.85 Million by 2033, growing at a CAGR of 9.9% during the forecast period 2025-2033.
The global achondroplasia market is undergoing a transformation driven by increasing awareness of rare genetic disorders, advancements in science, and growing demand for disease-modifying therapies. Achondroplasia, the most common form of dwarfism caused by mutations in the FGFR3 gene, has traditionally had limited treatment options. However, recent years have seen a shift towards targeted therapies, such as C-type natriuretic peptide (CNP) analogs like vosoritide, which have shown positive outcomes in pediatric patients.
Clinical trials are underway to assess the long-term safety and efficacy of emerging drugs, indicating a robust pipeline and strong market potential. Collaborations between biotech firms, academic institutions, and healthcare organizations are accelerating innovation and regulatory progress.
North America leads the market due to its advanced healthcare infrastructure, availability of approved drugs, strong patient advocacy networks, and supportive regulatory environment. Europe follows closely, supported by active research and early adoption of new therapies. The Achondroplasia market is expected to grow steadily, driven by technological innovation, supportive policies for orphan drug development, and the rising emphasis on precision medicine.
Global Achondroplasia Market: Executive Summary
Global Achondroplasia Market Dynamics: Drivers & Restraints
Driver: Advancements in targeted therapies
The global achondroplasia market is transforming due to advancements in targeted therapies. Achondroplasia, a rare genetic disorder caused by mutations in the FGFR3 gene, has limited therapeutic options. However, recent molecular and genetic research has led to the development of disease-specific treatments that directly target the underlying genetic pathways. One notable advancement is the introduction of C-type natriuretic peptide analogs, such as Vosoritide, which promote endochondral bone growth by counteracting the overactive FGFR3 signaling.
These drugs demonstrate improved efficacy in enhancing growth velocity and reducing the need for surgical intervention. Investigational therapies, including FGFR3 inhibitors and gene-editing platforms, are also gaining momentum. This shift towards precision medicine is expanding the treatment landscape, attracting significant investment, and offering hope for improved quality of life among patients with achondroplasia.
For instance, in September 2024, BridgeBio Pharma, a biopharmaceutical company focused on genetic diseases, was granted Breakthrough Therapy Designation by the FDA for oral infigratinib, a drug under development for children with achondroplasia. This designation expedites the development and regulatory review of drugs in the U.S. that meet rigorous criteria, demonstrating substantial improvement in clinically significant endpoints.
Driver: Rising awareness and early diagnosis
The global achondroplasia market is growing due to increased awareness and early diagnosis of the genetic condition. Public health education and rare disease advocacy are improving, leading to better understanding among healthcare professionals and the general public. This awareness has promoted early diagnosis through genetic testing and prenatal screening, enabling clinicians to identify the condition before birth or shortly after.
Early detection is crucial for timely intervention, such as growth monitoring and orthopedic planning. Improved diagnosis also leads to more accurate epidemiological data, enabling pharmaceutical companies and policymakers to estimate disease prevalence and plan targeted treatment strategies. As early diagnosis becomes more accessible, demand for innovative and specialized treatment options will continue to grow.
Restraint: Regulatory challenges
The global achondroplasia market faces significant regulatory challenges due to its rare genetic disorder. The development and approval of targeted therapies are complex and lengthy, with regulatory bodies like the FDA and EMA requiring comprehensive clinical data for pediatric patients. Trials must meet rigorous ethical and scientific standards, which can delay time-to-market and increase development costs. Challenges include recruiting adequate participants, post-marketing surveillance, risk management plans, and region-specific regulatory frameworks, which can discourage investment and slow innovation.
Opportunity: Expansion in emerging markets
The global achondroplasia market is experiencing significant growth in emerging markets, with the majority of advanced therapies and clinical developments concentrated in North America and Europe. These regions are experiencing rapid improvements in healthcare infrastructure, increased government spending on rare diseases, and rising awareness among healthcare professionals and the public. As diagnostic capabilities become more accessible and affordable, early identification of achondroplasia is likely to increase, driving demand for effective treatment options.
The rising middle-class population and improved insurance coverage in countries like India, China, and Brazil are also enhancing the affordability of specialty treatments. Pharmaceutical companies can leverage this opportunity by collaborating with local healthcare providers, launching educational campaigns, and establishing patient assistance programs.
For more details on this report, Request for Sample
Global Achondroplasia Market Segment Analysis
The global achondroplasia market is segmented based on drug type, route of administration, age group, distribution channel, and region.
Drug Type:
The CNP analogs segment from the drug type is expected to hold 43.1% of the achondroplasia market
The CNP analogs segment is a significant driver in the global achondroplasia market due to its targeted mechanism of action and promising clinical outcomes. Achondroplasia results from overactive signaling of the FGFR3 gene, inhibiting bone growth. C-type natriuretic peptide (CNP) analogs, like Vosoritide, counteract this effect by promoting endochondral ossification, stimulating bone growth in affected individuals.
The regulatory approval of Vosoritide in the U.S. and Europe has validated the CNP pathway as a therapeutic target and encouraged significant R&D investment. CNP analogs are non-invasive, making them easier for long-term pediatric use. The growing body of clinical evidence supporting their efficacy and safety, combined with strong demand for disease-modifying therapies, makes the CNP analogs segment a major growth driver in the global achondroplasia treatment landscape.
For instance, in June 2025, Ascendis Pharma received a priority review from the FDA for its New Drug Application for TransCon CNP (navepegritide) for treating children with achondroplasia. The FDA has set a PDUFA goal date of November 30, 2025, to complete the review. TransCon CNP is an investigational prodrug of C-type natriuretic peptide administered once weekly, designed to treat achondroplasia by providing continuous exposure of active CNP to receptors on various body tissues.
Global Achondroplasia Market - Geographical Analysis
North America dominated the global achondroplasia market with the highest share of 43.3% in 2024
North America dominates the Global Achondroplasia Market due to its strong healthcare infrastructure, high awareness, and early adoption of innovative therapies. Key players like BioMarin and Pfizer are investing in rare disease research, while regulatory support from the FDA accelerates targeted therapies. Advanced genetic testing, widespread insurance coverage, and patient advocacy groups facilitate early diagnosis and access to novel treatments, making North America a major growth hub for achondroplasia treatment innovations.
For instance, in March 2025, Ascendis Pharma submitted its New Drug Application (NDA) for TransCon CNP (navepegritide) to the U.S. Food & Drug Administration (FDA) for treating children with achondroplasia. TransCon CNP is an investigational prodrug of C-type natriuretic peptide (CNP) administered weekly, providing continuous exposure to active CNP receptors on growth plates and skeletal muscle.
Asia-Pacific is the global achondroplasia market with a market share of 21.5% in 2024
The Asia Pacific region is rapidly growing in the global achondroplasia market due to increased awareness, improved diagnostic capabilities, and healthcare investments. Countries like China, India, and Japan are enhancing medical infrastructure and genetic testing for early diagnosis and management of rare genetic disorders. Government initiatives and partnerships with international pharmaceutical firms are expanding access to advanced therapies. The region's large population and focus on rare diseases create a strong commercial opportunity for global players, making it a key region for future market expansion and innovation.
For instance, in February 2024, BridgeBio Pharma and Kyowa Kirin partnered to develop and commercialize infigratinib for achondroplasia, hypochondroplasia, and other skeletal dysplasias in Japan. BridgeBio's affiliate, QED Therapeutics, will grant Kyowa Kirin an exclusive license, with an upfront payment of $100 million and potential milestone-based payments. Infigratinib is an oral small molecule designed to target FGFR3-driven skeletal dysplasias at their source.
Global Achondroplasia Market - Key Players
The major global players in the achondroplasia market include BioMarin Pharmaceutical Inc., Ascendis Pharma A/S, Pfizer Inc., Roche Holding AG, Novartis AG, Ipsen S.A., OPKO Health Inc., QED Therapeutics, and LG Chem Life Sciences, among others.
Global Achondroplasia Market – Key Developments
In May 2025, BioMarin Pharmaceutical Inc. released new data from studies on VOXZOGO, a drug that has shown significant impact on tibial bowing in children with achondroplasia and has shown early efficacy in other skeletal conditions. The data was presented at the 2025 Joint Congress of the European Society for Paediatric Endocrinology and the European Society of Endocrinology in Denmark.
Global Achondroplasia Market: Scope
Metrics | Details | |
CAGR | 9.9% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Mn) | |
Segments Covered | Type | CNP Analogs (Vosoritide), FGFR3 Inhibitors, Growth Hormone Therapies |
Route of Administration | Oral, Injectable | |
| Age Group | Pediatric, Adult |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
DMI Insights:
The global achondroplasia market is experiencing rapid growth due to targeted therapies like CNP analogs and FGFR3 inhibitors. The market is projected to grow at a CAGR of 9.9% from 2025 to 2033, driven by a shift towards precision medicine and disease-modifying treatments. North America leads with robust R&D and regulatory support, while emerging markets are becoming attractive expansion zones due to increased awareness and healthcare infrastructure.
The global achondroplasia market report delivers a detailed analysis with 67 key tables, more than 59 visually impactful figures, and 178 pages of expert insights, providing a complete view of the market landscape.
Suggestions for Related Report
For more pharmaceutical-related reports, please click here
























































