Market Overview
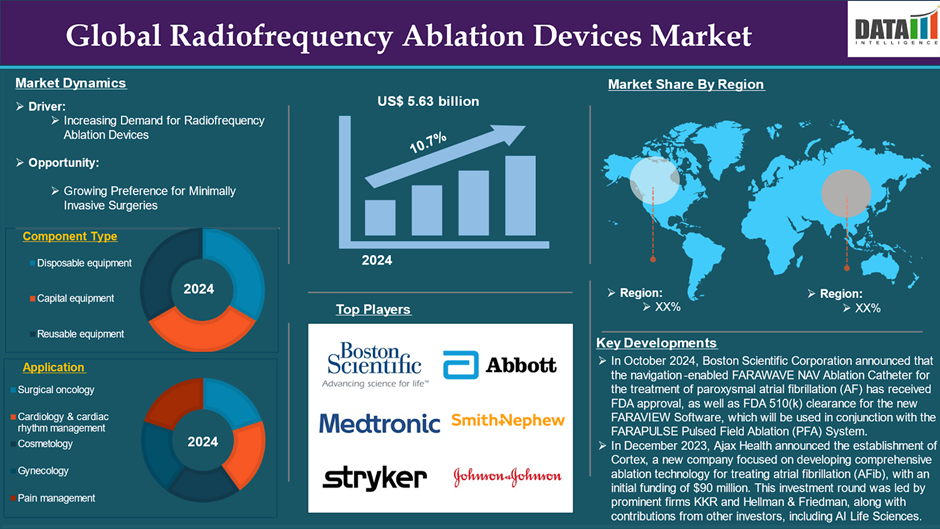
The Global Radiofrequency Ablation Devices Market reached US$ 5.63 billion in 2024 and is expected to reach US$ 13.54 billion by 2033, growing at a CAGR of 10.7% during the forecast period 2025-2033.
Radiofrequency ablation (RFA) is a minimally invasive medical procedure that generates heat from high-frequency alternating current and then uses it to eliminate or ablate certain tissues in the body. This procedure is widely used in a variety of medical specialties, including interventional radiology and cardiology, and is especially helpful in treating tumors, cardiac arrhythmias, and chronic pain. The method involves introducing a tiny needle or catheter into the desired location and applying radiofrequency radiation to induce localized heating. This heat causes the destruction of aberrant tissue, diminishing or eliminating its function.
RFA has a wide variety of applications. In oncology, it is used to destroy malignancies in organs like the liver, lungs, and kidneys. In cardiology, RFA is used to address faulty electrical circuits in the heart that cause arrhythmia. It is also used in pain management to disrupt nerve signals that cause chronic pain diseases including arthritis and lower back pain. RFA's versatility makes it an essential instrument in modern medicine, providing patients with superior healthcare alternatives that are minimally intrusive.
Executive Summary
For more details on this report – Request for Sample
Market Dynamics: Drivers & Restraints
Increasing demand for radiofrequency ablation devices
The increasing demand for radiofrequency ablation devices is expected to be a significant factor in the growth of the global radiofrequency ablation devices market. The demand for on-body drug delivery devices is increasing owing to the rising prevalence of chronic diseases, technological advancements, growing preference for minimally invasive surgical procedures, and recent launches and approvals.
As chronic disorders like cancer, cardiovascular disease, and chronic pain become more common, healthcare providers are looking for efficient treatment alternatives that will reduce patient misery and recovery time. For instance, according to the American College of Cardiology Foundation, High systolic blood pressure contributed the most to age-standardized CVD disability-adjusted life years (DALYs), with 2,564.9 per 100,000 people worldwide.
RFA is an enticing solution because it enables precision targeting of aberrant tissues while maintaining adjacent healthy structures. This feature is especially useful in oncology, where RFA is used to treat solid tumors, giving it a viable option for patients who may not be candidates for standard surgery. The market is also driven by technological advancements that improve the efficacy, safety, and usability of these devices in a variety of medical settings. As the healthcare landscape changes, advances in RFA technology make these devices more accessible and effective for treating a wide range of ailments, including chronic diseases and malignancies, which are becoming more common worldwide. One of the most significant advances in RFA technology has been the introduction of multi-needle electrode devices. These methods enable more precise tissue targeting, allowing doctors to treat larger areas with higher accuracy while reducing damage to healthy tissues nearby. This accuracy is critical in oncology, where the ability to properly ablate tumor cells while conserving healthy structures has a substantial impact on patient outcomes.
Furthermore, the inclusion of real-time monitoring capabilities in RFA devices gives clinicians immediate feedback during operations. This feature provides greater control over the energy delivery process, ensuring that the proper temperature is maintained throughout the treatment, improving both safety and effectiveness. There are recent launches and approvals of radiofrequency ablation devices. For instance, in October 2024, Boston Scientific Corporation announced that the navigation-enabled FARAWAVE NAV Ablation Catheter for the treatment of paroxysmal atrial fibrillation (AF) has received FDA approval, as well as FDA 510(k) clearance for the new FARAVIEW Software, which will be used in conjunction with the FARAPULSE Pulsed Field Ablation (PFA) System to provide visualization for cardiac ablation procedures. These technologies are only compatible with Boston Scientific's existing cardiac mapping technology and the company's newest product, the OPAL HDx Mapping System.
Side effects and complications associated with radiofrequency ablation devices
Factors such as side effects and complications associated with radiofrequency ablation devices are expected to hamper the global radiofrequency ablation devices market. RFA is generally regarded as a safe and minimally intrusive therapy, however, it is not without potential hazards. Patients undergoing RFA may have a variety of side effects, including discomfort, edema, and hypersensitivity near the injection site. These symptoms might emerge as burning sensations or numbness, similar to sunburn, and may last for days or even weeks after the surgery. Such discomfort may cause patient discontent and reluctance to pursue this treatment in the future, affecting overall market acceptance and growth. Furthermore, more significant problems, albeit uncommon, can arise during or following the RFA operation. Infections at the injection site, nerve and blood vessel injury, and bleeding are all possible complications.
Market Segment Analysis
The global radiofrequency ablation devices market is segmented based on component type, application, end-user, and region.
Capital equipment segment is expected to dominate the global radiofrequency ablation devices market share
The capital equipment segment is anticipated to dominate the global radiofrequency ablation devices market owing to its increasing prevalence of chronic diseases, growing need for effective treatment options, technological advancements, and recent launches and approvals. This expansion relies heavily on capital equipment, which includes expensive goods such as radiofrequency generators and sophisticated ablation devices. These technologies improve both therapy efficacy and patient outcomes, making them indispensable in modern medical procedures. Furthermore, the capital equipment segment benefits from the continuing trend of technological innovation in healthcare. As hospitals and clinics invest in modern RFA systems, they become better prepared to manage complex cases requiring precision and dependability.
Technological advancements in RFA devices have resulted in increased precision and control during operations. Modern RFA systems use modern electrosurgical generators to deliver heat energy more precisely, allowing for targeted tissue destruction while protecting surrounding healthy tissue. These advances not only improve treatment outcomes but also cut recovery times, making RFA a more compelling alternative for patients. There are recent launches and approvals which are manufactured using advanced technology.
For instance, in October 2024, Abbott announced that it has completed two major milestones to support the company's growing suite of pulsed-field ablation (PFA) solutions in electrophysiology, early enrollment in the VOLT-AF IDE Study supporting the Volt PFA System, and the launch of the FOCALFLEX trial to assess the company's TactiFlex Duo Ablation Catheter, Sensor Enabled (SE), which will be used in the treatment of patients with paroxysmal atrial fibrillation. These two trials offer major benefits for the future of two Abbott PFA catheters, which are being developed as critical tools for clinicians treating patients with irregular heart rhythms like as atrial fibrillation (AFib).
Market Geographical Share
North America is expected to hold a significant position in the global radiofrequency ablation devices market share
North America will likely account for a significant share of the global radiofrequency ablation devices market, owing to its advanced healthcare infrastructure, increasing investments, a high prevalence of chronic diseases, technological advancements, and recent launches and approvals. This upward trend is primarily attributable to the increasing number of people suffering from chronic ailments such as cancer, arthritis, and cardiovascular disease.
For instance, according to the National Institute of Health, approximately 82.6 million people in the United States suffer from one or more forms of cardiovascular disease (CVD). Coronary heart disease (CHD), stroke, hypertension, and congestive heart failure are among the most common forms of cardiovascular disease. CHD affects an estimated 16.3 million Americans aged 20 and older, accounting for a 7% prevalence. The prevalence for men is 8.3 percent, while for women it is 6.1 percent. About 7 million Americans aged 20 and up have experienced a stroke. Each year, around 610,000 people have their first stroke, with another 185,000 experiencing a recurrence. Approximately 87 percent of all strokes are ischemic, with 10% caused by intracerebral hemorrhage and 3% by subarachnoid hemorrhage. Cardiovascular disease, diabetes, and autoimmune disorders are becoming more prevalent in the population. This frightening trend emphasizes the critical need for improved medical technologies capable of providing appropriate therapies for individuals suffering from chronic diseases.
The development of multi-needle electrodes and enhanced visualization techniques is one of the important technological advances that is driving the market. These advancements give more precision during surgeries, allowing medical professionals to target specific locations more effectively while limiting injury to neighboring tissues. Real-time imaging integration improves the visibility of anatomical features, which is critical for successful therapies. As a result, patients experience better outcomes and shorter recovery times, making RFA a better choice for both physicians and patients.
The market expansion is being driven by increasing expenditures on healthcare and a determined push to adopt modern medical technology, which is critical for tackling the growing prevalence of chronic diseases such as cancer and cardiovascular ailments.
The increased recognition of the benefits of radiofrequency ablation techniques is a major driver of this investment boom. As healthcare providers strive to improve patient outcomes while reducing costs, RFA technology investments have become a top priority. Hospitals and clinics are rapidly spending money to acquire cutting-edge RFA devices with improved precision and safety features. For instance, in June 2024, Kardium, the developer of a pulsed field ablation and mapping system for atrial fibrillation (AFib), announced that it has raised $104 million in the latest fundraising round to assist the firm prepare for commercialization. The funds will be used to finish Kardium's Pulsar clinical study, which is required to secure regulatory approval for their Globe mapping and ablation device. In addition, the company intends to increase its production team and capacity, as well as establish clinical support and commercial teams.
Asia Pacific is growing at the fastest pace in the global radiofrequency ablation devices market
The Asia Pacific region is emerging as the fastest-growing market for radiofrequency ablation devices, owing to a combination of demographic trends, increased healthcare investment, technological advancements, recent launches, and a rising number of chronic diseases. This increase is largely due to the increased prevalence of disorders like as cardiovascular disease (CVDs) and cancer, which require effective treatment alternatives such as RFA.
For instance, according to the National Institute of Health, Coronary heart disease prevalence rates in India have been calculated throughout the last several decades, ranging from 1.6% to 7.4% in rural populations and 1% to 13.2% in metropolitan areas. This disturbing development highlights the critical need for modern medical therapies, such as RFA, to successfully manage these chronic illnesses. The rising prevalence of chronic diseases in Asia Pacific is exacerbated by demographic changes, notably an aging population.
Competitive Landscape
The major global players in the global radiofrequency ablation devices market include Boston Scientific Corporation, Medtronic, Stryke, Abbott, Smith+Nephew, Johnson & Johnson Services, Inc., AngioDynamic, Hologic, Inc., Arthrex, Inc., STARmed America among others.
Emerging Players
Ananya Health, Sedivention, and Field Medical among others
| Metrics | Details | |
| CAGR | 4.5% | |
| Market Size Available for Years | 2018-2033 | |
| Estimation Forecast Period | 2025-2033 | |
| Revenue Units | Value (US$ Mn) | |
| Segments Covered | Component Type | Disposable equipment, Capital equipment, Reusable equipment |
| Application | Surgical oncology, Cardiology & cardiac rhythm management, Cosmetology, Gynecology, Pain management | |
| End-User | Hospitals, Ambulatory Surgical Centers, Others | |
| Regions Covered | North America, Europe, Asia-Pacific, South America, and Middle East & Africa | |
Key Developments
In December 2023, Ajax Health announced the establishment of Cortex, a new company focused on developing comprehensive ablation technology for treating atrial fibrillation (AFib), with an initial funding of $90 million. This investment round was led by prominent firms KKR and Hellman & Friedman, along with contributions from other investors, including AI Life Sciences. The funding will support Cortex's mission to create advanced diagnostic and pulsed-field ablation (PFA) catheters, which are essential for improving treatment outcomes for patients suffering from AFib.
Why Purchase the Report?
- Pipeline & Innovations: Reviews ongoing clinical trials, product pipelines, and forecasts upcoming advancements in medical devices and pharmaceuticals.
- Product Performance & Market Positioning: Analyzes product performance, market positioning, and growth potential to optimize strategies.
- Real-World Evidence: Integrates patient feedback and data into product development for improved outcomes.
- Physician Preferences & Health System Impact: Examines healthcare provider behaviors and the impact of health system mergers on adoption strategies.
- Market Updates & Industry Changes: Covers recent regulatory changes, new policies, and emerging technologies.
- Competitive Strategies: Analyzes competitor strategies, market share, and emerging players.
- Pricing & Market Access: Reviews pricing models, reimbursement trends, and market access strategies.
- Market Entry & Expansion: Identifies optimal strategies for entering new markets and partnerships.
- Regional Growth & Investment: Highlights high-growth regions and investment opportunities.
- Supply Chain Optimization: Assesses supply chain risks and distribution strategies for efficient product delivery.
- Sustainability & Regulatory Impact: Focuses on eco-friendly practices and evolving regulations in healthcare.
- Post-market Surveillance: Uses post-market data to enhance product safety and access.
- Pharmacoeconomics & Value-Based Pricing: Analyzes the shift to value-based pricing and data-driven decision-making in R&D.
The global radiofrequency ablation devices market report delivers a detailed analysis with 60+ key tables, more than 50 visually impactful figures, and 176 pages of expert insights, providing a complete view of the market landscape.
Target Audience 2025
- Manufacturers: Pharmaceutical, Medical Device, Biotech Companies, Contract Manufacturers, Distributors, Hospitals.
- Regulatory & Policy: Compliance Officers, Government, Health Economists, Market Access Specialists.
- Technology & Innovation: AI/Robotics Providers, R&D Professionals, Clinical Trial Managers, Pharmacovigilance Experts.
- Investors: Healthcare Investors, Venture Fund Investors, Pharma Marketing & Sales.
- Consulting & Advisory: Healthcare Consultants, Industry Associations, Analysts.
- Supply Chain: Distribution and Supply Chain Managers.
- Consumers & Advocacy: Patients, Advocacy Groups, Insurance Companies.
- Academic & Research: Academic Institutions.
























































