Global Certified Reference Materials Market Size
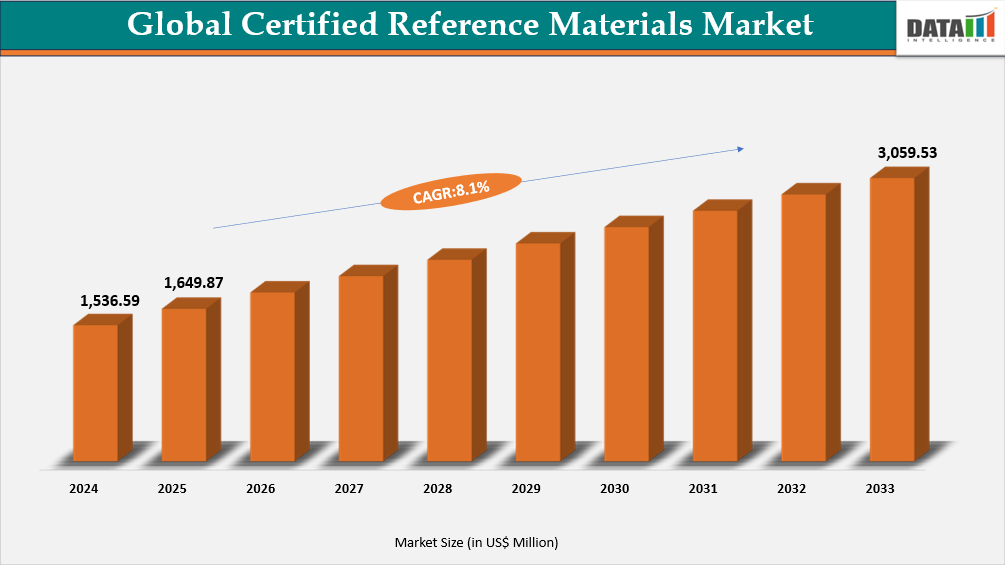
Global Certified Reference Materials Market reached US$ 1,649.87 Million in 2025 and is expected to reach US$ 3,059.53 Million by 2033, growing with a CAGR of 8.1% during the forecast period 2026-2033.
The certified reference materials (CRM) market is experiencing robust global growth, driven by increasing regulatory scrutiny and demand for high-precision testing across pharmaceuticals, food safety, environmental monitoring, and advanced manufacturing. Adoption of ultra-sensitive analytical technologies such as ICP-MS and LC-MS/MS is expanding CRM use into trace-level detection for semiconductors, biologics, and advanced materials.
In India, government initiatives like PMKSY and FSSAI’s lab expansion are significantly boosting CRM deployment in food quality and environmental monitoring. In the US and Europe, tightening chemical safety regulations and environmental programs are sustaining demand for matrix-matched CRMs in public health, manufacturing, and pharmaceuticals. China’s expanding ingredient databases and cosmetic testing programs, along with Brazil’s focus on food export compliance and environmental oversight, further reinforce global CRM adoption. Overall, CRMs are increasingly critical not just for calibration, but for regulatory compliance and precision-driven analytical applications worldwide.
Certified Reference Materials Industry Trends and Strategic Insights
- Asia-Pacific is the fastest-growing region in the certified reference materials market, capturing the share of 35.4% in 2025.
- By end-user industry, the environmental testing is projected to be the largest market, holding a significant share of about 23% in 2025.
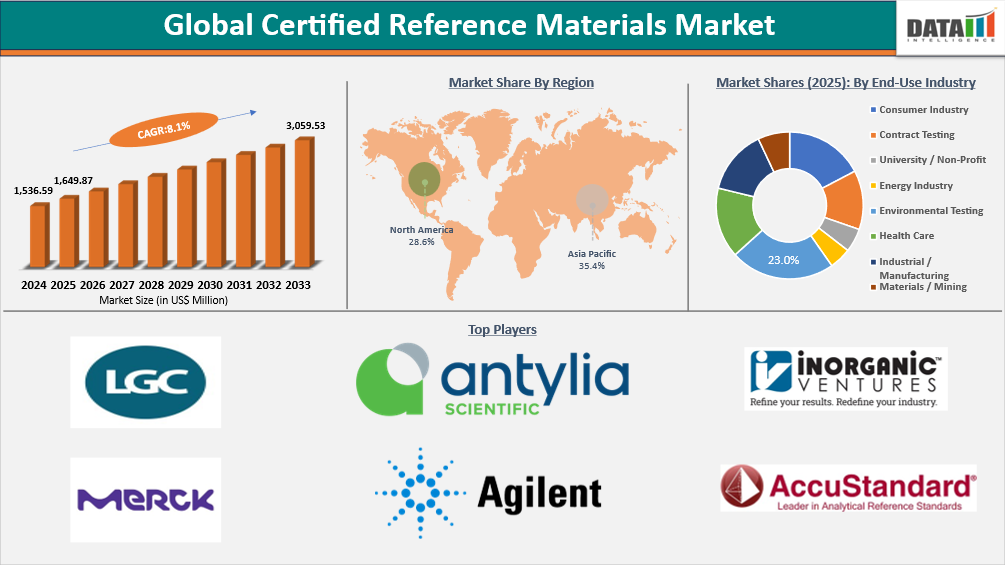
Global Certified Reference Materials Market Size and Future Outlook
- 2025 Market Size: US$ 1,649.87 Million
- 2033 Projected Market Size: US$ 3,059.53 Million
- CAGR (2026-2033): 8.1%
- Largest Market: Asia-Pacific
- Fastest Market: Asia-Pacific
Market Scope
| Metrics | Details |
| By Product Type | Organic, Inorganic |
| By Application | Quality Control & Assurance, Method Validation, Regulatory Compliance Testing, R&D & Analytical Development, Calibration & Instrument Validation |
| By End-User Industry | Consumer Industry, Contract Testing, University / Non-Profit, Energy Industry, Environmental Testing, Health Care, Industrial / Manufacturing, Materials / Mining |
| By Region | North America, Latin America, Europe, Asia-Pacific, Middle East and Africa |
| Report Insights Covered | Competitive Landscape Analysis, Company Profile Analysis, Market Size, Share, Growth |
For more Detail information, Request for Sample
Market Dynamics
Stricter Global Contaminant Limits Driving Ultra-Trace CRM Demand
Tightening global regulations on contaminants are significantly increasing demand for ultra-trace certified reference materials (CRMs) across food safety, environmental testing, pharmaceuticals, and industrial applications. Frameworks such as Regulation (EU) 2023/915, standards from the U.S. Food and Drug Administration, and guidelines by the Codex Alimentarius Commission are lowering permissible limits for pesticides, heavy metals, and toxins to parts-per-billion and parts-per-trillion levels. This shift requires highly precise and traceable CRMs to ensure analytical accuracy and regulatory compliance.
The impact is especially pronounced in food and agriculture, where stringent import/export requirements demand validated proof of compliance with contaminant limits. This has accelerated adoption of advanced analytical technologies such as LC-MS and GC-MS, which depend on high-purity, matrix-matched CRMs for calibration and validation. As a result, laboratories are increasingly relying on specialized CRMs that replicate real sample compositions to accurately detect ultra-low contaminant concentrations, reinforcing sustained market growth.
High Cost and Technical Complexity of ISO 17034 Accreditation
A major constraint in the global Certified Reference Materials market is the high cost and technical complexity of obtaining and maintaining ISO 17034 accreditation, which specifies the competence requirements for reference material producers. Unlike conventional laboratory certifications, this standard requires strict control over the complete production process, including material characterization, homogeneity testing, stability assessment, uncertainty evaluation, and traceability to the International System of Units. Meeting these requirements demands advanced metrological instruments, specialized technical expertise, and highly qualified scientific personnel, significantly increasing operational costs for CRM producers.
The accreditation process is also time-intensive and resource-demanding. Producers must implement strong quality management systems aligned with ISO 17034 and ensure analytical laboratories used for CRM characterization comply with ISO/IEC 17025. Certification involves extensive documentation audits, proficiency testing, inter laboratory validation studies, and continuous surveillance by accreditation bodies. Due to these financial and technical barriers, the CRM market is largely dominated by established players, while smaller or emerging manufacturers face difficulties entering the market, particularly in developing regions.
Segmentation Analysis
The global certified reference materials market is segmented based on product type, application, end-user industry, and region.
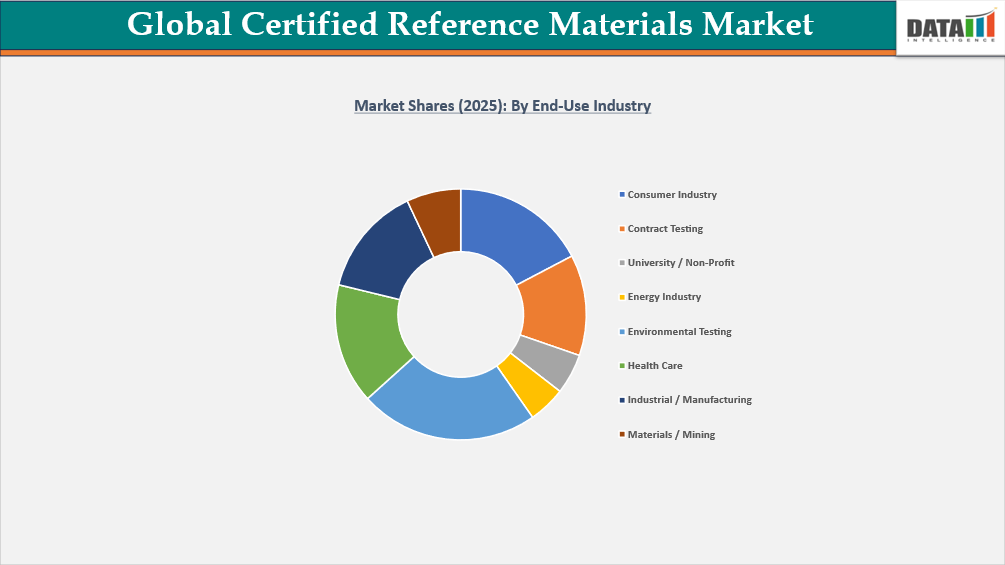
Stringent Regulatory Compliance and Expanding Monitoring Programs Fuel Environmental Testing CRM Market Growth
The Environmental Testing segment constitutes one of the most expansive and structurally anchored demand centers within the global CRM market, with 23% market share, driven by an unrelenting cascade of regulatory compliance requirements, climate-linked monitoring mandates, and the proliferation of environmental surveillance programs administered at national and supranational levels. From water quality verification to soil contamination assessment and ambient air monitoring, environmental testing laboratories operate under some of the most stringent metrological requirements in applied analytical science, which place certified reference materials at the center of every credible measurement workflow.
Water quality monitoring represents the largest single application category within environmental testing CRM demand. The U.S. EPA administers the Safe Drinking Water Act, which currently regulates 90 contaminants in public water systems. The EPA's National Primary Drinking Water Regulations establish maximum contaminant levels (MCLs) for inorganic chemicals, organic compounds, and microbiological contaminants, each category requiring CRM-calibrated analytical measurement for compliance verification. Ambient air quality monitoring requires a distinct portfolio of certified gas mixture standards and particulate reference materials. The U.S. EPA's National Ambient Air Quality Standards (NAAQS) establish limits for six criteria pollutants: particulate matter (PM2.5 and PM10), ozone, nitrogen dioxide, sulfur dioxide, carbon monoxide, and lead.
Precision Medicine and Compliance Requirements Fuel Growth in Healthcare CRM Segment
The Healthcare segment stands as one of the most analytically rigorous and metrologically demanding domains of CRM consumption, encompassing in vitro diagnostics (IVD), clinical laboratory testing, pharmaceutical quality control, medical device calibration, and biomarker research. Patient safety imperatives, regulatory mandates enforced by bodies including the FDA, EMA, and WHO, and the normative infrastructure of international standards for in vitro diagnostics collectively position CRM use in healthcare as both a regulatory obligation and a fundamental quality assurance requirement. The convergence of precision medicine, pandemic-driven diagnostic capacity expansion, and increasingly stringent global laboratory regulations is structurally expanding healthcare CRM demand.
In vitro diagnostics represent the largest and most systematized application of CRMs within the healthcare segment. ISO 17511, which establishes metrological traceability of values assigned to calibrators and control materials in in vitro diagnostic medical devices, mandates that assigned values be traceable to higher-order reference measurement procedures or reference materials requirements fulfilled by commutable CRMs. The CDC's CRMLN currently comprises designated comparison method laboratories in multiple countries, each serving as a traceability anchor for commercial IVD manufacturers.
Geographical Penetration
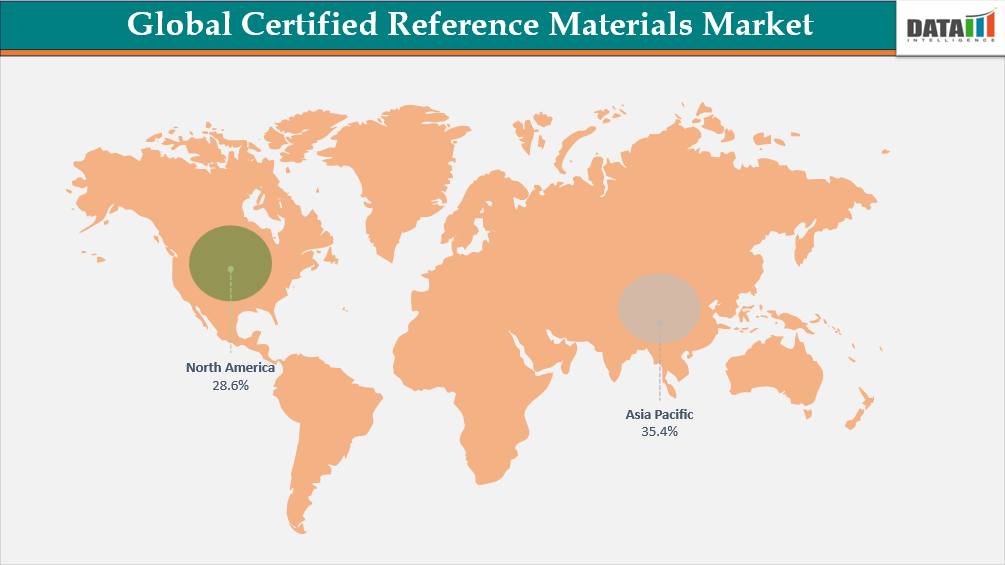
Government Initiatives and Rising Analytical Demand Drive Asia-Pacific Certified Reference Materials Growth
Asia-Pacific Certified Reference Materials (CRM) market is witnessing accelerated growth, driven by expanding regulatory oversight, the maturation of metrology infrastructure, and increasing demand for traceability across analytical laboratories. As economies across the region industrialize and diversify, the need for reliable calibration standards and analytical benchmarks has become central to quality assurance in pharmaceuticals, food safety, environmental monitoring, clinical diagnostics, and industrial chemistry. Countries like China, Japan, South Korea, India, and Australia have significantly increased their domestic CRM production capacity, while Southeast Asian nations are enhancing their reliance on accredited reference materials through international collaboration.
Government-led initiatives are a major catalyst. For instance, NIM offers 2502 certified reference materials (CRMs), 1121 primary reference materials, and 1381 secondary reference materials, which cover 11 areas. Out of these, about 27% is for environmental contaminants such as heavy metals and persistent organic pollutants, aligning with its “Green China” policy and updated ambient air and water standards. Asia-Pacific CRM market continues to mature as regional regulatory and accrediting infrastructures improve. In 2024, Asia-Pacific-affiliated accreditation bodies accredited over 75,000 laboratories for reference materials (ISO 17034), calibration, testing, and more. This shows a significant increase in regional capacity for high-quality CRM development and certification. This institutional strengthening promotes better consistency in CRM quality, hence facilitating cross-border acceptance under both the Asia-Pacific and ILAC mutual recognition agreements.
China Certified Reference Materials Market Trends
China’s certified reference materials (CRM) market is expanding steadily, supported by strong regulatory oversight, large-scale testing programs, and continuous enhancement of national reference databases. Increasing focus on product safety, particularly in pharmaceuticals, cosmetics, and food, is driving demand for high-quality CRMs to ensure traceability, validation, and compliance across analytical laboratories. The country’s growing metrology infrastructure and emphasis on domestic standardization are further strengthening CRM adoption across industrial and healthcare applications.
In 2025, the National Institutes for Food and Drug Control (NIFDC) updated the Ingredient Usage Information for Marketed Products database, expanding from approximately 2,200 ingredient types in 2024 to 3,000 in 2025, enhancing reference data for safety assessments. Additionally, in 2025, China’s National Medical Products Administration (NMPA) tested over 21,000 cosmetic batches across multiple product categories through 33 institutions, reinforcing demand for CRM-supported validation and compliance-driven analytical workflows.
India Certified Reference Materials Market Outlook
India’s certified reference materials (CRM) market is witnessing strong growth, driven by expanding regulatory enforcement, rising laboratory accreditation, and increasing demand for traceability across pharmaceuticals, food safety, environmental testing, and industrial applications. Government-led initiatives such as CSIR–NPL’s Bharatiya Nirdeshak Dravya (BND) program are strengthening domestic CRM production and reducing reliance on imports, aligning with the country’s broader self-reliance and quality infrastructure goals. Growing NABL accreditation under ISO 17034 and the rapid expansion of testing laboratories are further accelerating CRM adoption across sectors.
In November 2025, the Department of Atomic Energy, an India-based government research and nuclear science organization, launched an indigenously developed certified reference material (CRM) titled Ferrocarbonatite (FC)-BARC B1401 to support rare earth element (REE) exploration, analytical calibration, and quality control across mining, electronics, and advanced materials sectors. This development highlights India’s increasing capability in high-value CRM production and its strategic focus on supporting advanced manufacturing and resource-driven industries.
Product and Portfolio Analysis
Routine Testing Dependence and Specialized Applications Drive Dual Demand for Stock and Custom CRMs
Globally, laboratories continue to rely heavily on stock Certified Reference Materials to support routine analytical workflows. For example, ISO/IEC 17025 accreditation has been adopted by more than 114,600 laboratories worldwide, many of which perform daily or weekly calibration checks using ready-to-use standards to ensure measurement validity. In environmental testing programs alone, public water systems in US conduct monitoring for contaminants such as lead and arsenic across more than 150,000 regulated systems, often on monthly or quarterly schedules, reinforcing the need for standardized stock materials to maintain consistency across sampling cycles.
At the same time, the use of customized CRMs is growing in applications where analytical conditions are more complex. Under pharmaceutical guidelines such as ICH Q3D, manufacturers may validate impurity testing methods across multiple dosage forms, requiring product-specific standards to match formulation matrices. Similarly, in advanced manufacturing environments such as semiconductor fabrication, contamination monitoring at sub-parts-per-billion levels requires matrix-matched reference materials rather than generic solutions. This indicates a clear global operating pattern: stock CRMs are essential for high-frequency routine testing across thousands of regulated laboratories, while custom CRMs are increasingly deployed in specialized applications where method specificity and matrix alignment are critical.
Customer Buying Behavior Analysis
Supply Reliability, Quality Consistency, and Evolving Analytical Needs Drive CRM Supplier Switching Behavior
Laboratories typically maintain long-term relationships with CRM suppliers due to the importance of measurement consistency and regulatory compliance. However, certain conditions can trigger supplier switching. One of the most common triggers is persistent supply disruptions or long delivery times, particularly when laboratories require frequently used standards for routine testing workflows. Quality issues also represent a major switching factor. If laboratories experience inconsistencies in CRM certification data, stability performance, or analyte concentrations, they may evaluate alternative suppliers. Because CRM performance directly affects analytical accuracy, laboratories cannot risk using materials that compromise measurement reliability.
Pricing changes can also influence supplier switching decisions, particularly for laboratories with high CRM consumption. Contract testing laboratories that purchase large volumes of standards often periodically review supplier pricing and may shift to alternative providers offering competitive pricing structures or bulk discounts. Emerging analytical requirements can also trigger supplier changes. As laboratories adopt new testing methods, for example, PFAS contamination analysis or new pharmaceutical impurity testing protocols, they may seek suppliers capable of producing specialized or custom reference materials that are not available from their existing suppliers.
Regulatory Analysis
Global regulatory frameworks are significantly intensifying the demand for certified reference materials (CRMs) across food safety and environmental testing domains. In Europe, regulations such as EU 2023/915 on food contaminants and the Water Framework Directive are enforcing stricter limits on heavy metals, mycotoxins, and emerging pollutants like PFAS, driving adoption of matrix-specific and ultra-trace CRMs. Similarly, in the U.S., the EPA’s PFAS Action Plan and Method 1633, along with the FDA’s Food Safety Modernization Act (FSMA), are mandating standardized contaminant monitoring, accelerating the use of PFAS-specific, pesticide, and food contaminant reference materials.
In Asia, China’s Environmental Protection and Soil Pollution laws are strengthening monitoring requirements for heavy metals and pollutants, increasing demand for inorganic and matrix CRMs. At a global level, Codex Alimentarius standards and ICH guidelines are reinforcing the need for multi-residue pesticide standards and pharmaceutical impurity reference materials to support international trade and drug safety. Additionally, ISO/IEC 17025 accreditation requirements continue to anchor CRM demand across laboratories worldwide by mandating traceability and validated analytical methods.
Competitive Landscape
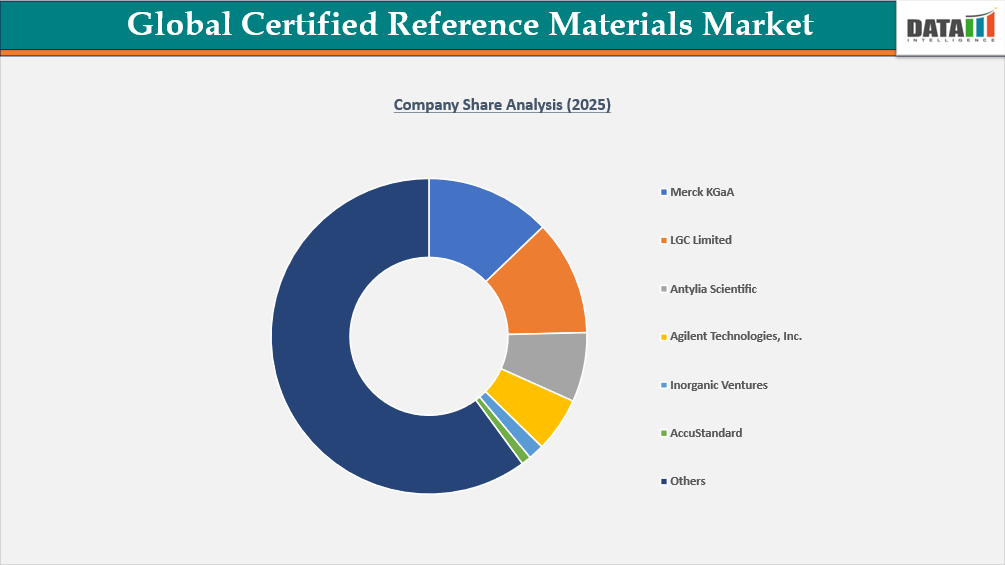
- The global certified reference materials market is characterized by a competitive landscape that includes both established and regional players.
- Key players include LGC Limited, AccuStandard, Inorganic Ventures, Antylia Scientific, Merck KGaA, CPAChem, AnalytiChem Holding GmbH, Starna Scientific Limited, FLUXANA GmbH & Co. KG, and Alpha Resources LLC.
Key Developments
- In February 2026, LGC Limited, a UK-based life sciences and analytical solutions company, expanded its reference materials portfolio and North American presence through the acquisition of the ChromaDex Reference Standards business from Niagen Bioscience, strengthening its capabilities in phytochemical standards and enhancing support for pharmaceutical, food, and testing laboratories.
- In March 2025, AccuStandard launched a new certified reference material (CRM) for PFAS analysis, including all 29 target compounds specified under the EN 17892 method, to support compliance with EU drinking water regulations and enable accurate detection of persistent contaminants.
Why Choose DataM?
- Data-Driven Insights: Dive into detailed analyses with granular insights such as pricing, market shares and value chain evaluations, enriched by interviews with industry leaders and disruptors.
- Post-Purchase Support and Expert Analyst Consultations: As a valued client, gain direct access to our expert analysts for personalized advice and strategic guidance, tailored to your specific needs and challenges.
- White Papers and Case Studies: Benefit quarterly from our in-depth studies related to your purchased titles, tailored to refine your operational and marketing strategies for maximum impact.
- Annual Updates on Purchased Reports: As an existing customer, enjoy the privilege of annual updates to your reports, ensuring you stay abreast of the latest market insights and technological advancements. Terms and conditions apply.
- Specialized Focus on Emerging Markets: DataM differentiates itself by delivering in-depth, specialized insights specifically for emerging markets, rather than offering generalized geographic overviews. This approach equips our clients with a nuanced understanding and actionable intelligence that are essential for navigating and succeeding in high-growth regions.
- Value of DataM Reports: Our reports offer specialized insights tailored to the latest trends and specific business inquiries. This personalized approach provides a deeper, strategic perspective, ensuring you receive the precise information necessary to make informed decisions. These insights complement and go beyond what is typically available in generic databases.
Target Audience 2026
- Manufacturers/ Buyers
- Industry Investors/Investment Bankers
- Research Professionals
- Emerging Companies