Biosimilars Market Overview
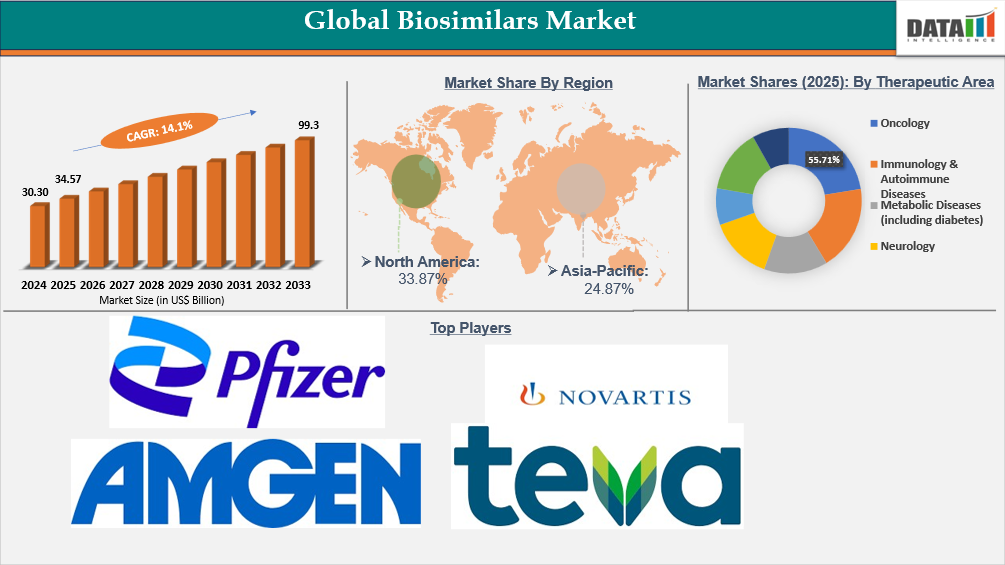
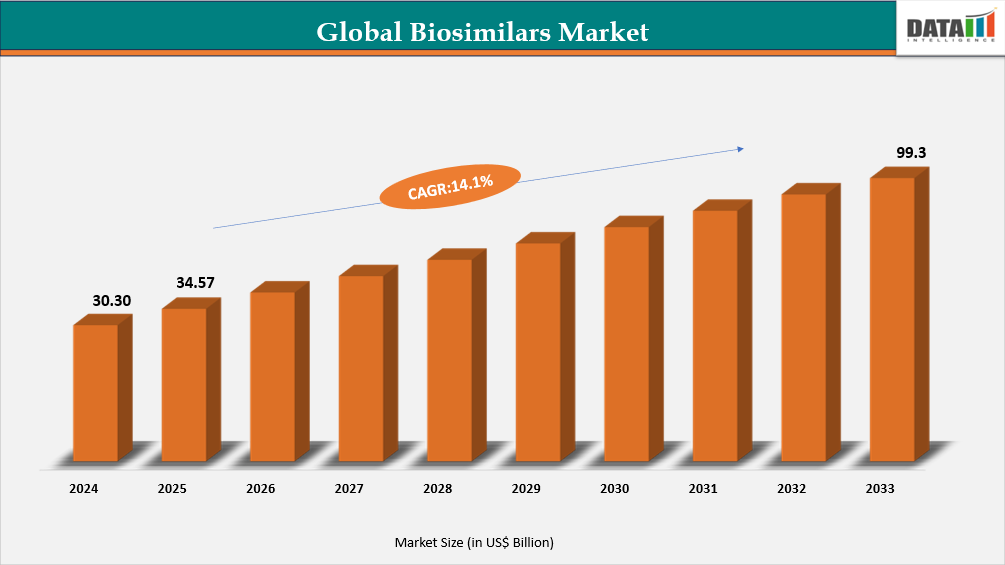
Global biosimilars market reached US$30.30 Billion in 2024, rising to US$ 34.57 Billion in 2025 and is expected to reach US$99.3 Billion by 2033, growing at a CAGR of 14.1% from 2026 to 2033.
The biosimilars market is expanding rapidly as healthcare systems seek to control the rising cost of biologic therapies while maintaining treatment access. Biologic medicines account for a significant share of global pharmaceutical spending, and in several developed markets they represent more than one-third of total drug expenditure, creating strong incentives for biosimilar substitution. Over 40 biosimilars have been approved in the U.S. alone, reflecting increasing regulatory confidence and clinical acceptance. Growing patent expiries of blockbuster biologics in oncology, immunology, and diabetes continue to open new opportunities for biosimilar developers.
Demand is also supported by expanding manufacturing capacity and increasing investments in biologics production infrastructure worldwide. More than 300 biosimilar candidates are currently reported to be in various stages of clinical development globally, indicating a robust future pipeline. Emerging markets in Asia are strengthening domestic biologics manufacturing, improving supply availability and lowering treatment costs. Increasing physician familiarity, supportive reimbursement frameworks, and broader inclusion of biosimilars in treatment guidelines are collectively accelerating global adoption across hospital and specialty care settings.
Biosimilars Market Industry Trends and Strategic Insights
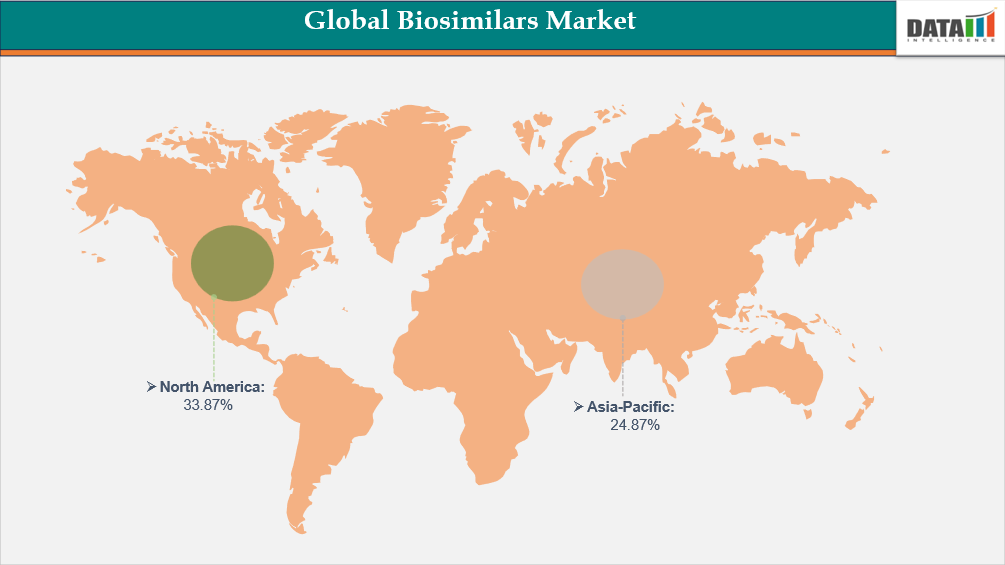
- North America leads the global biosimilars market, capturing the largest revenue share of 33.87% in 2025.
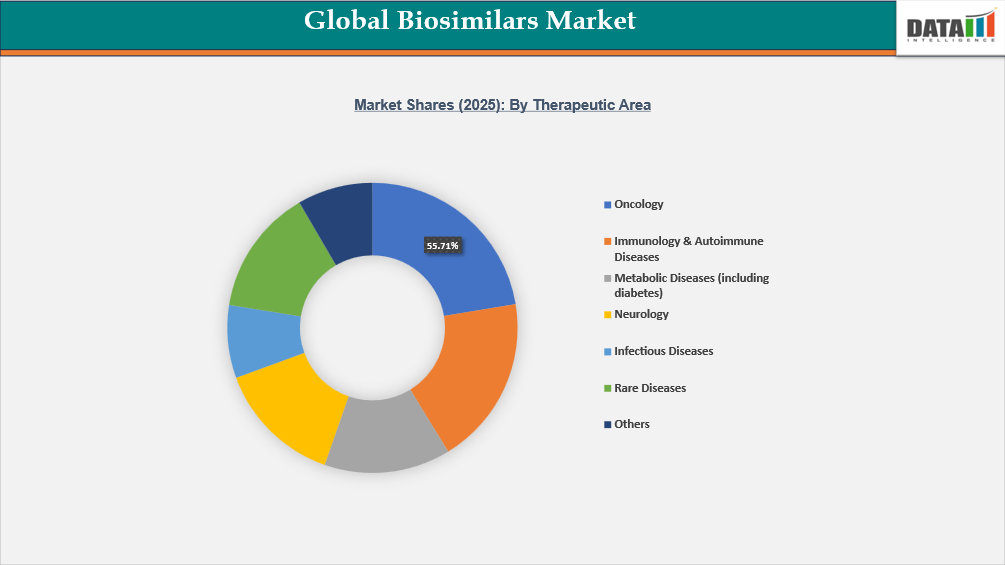
- By therapeutic area segment, oncology led the global biosimilars market, capturing the largest revenue share of 55.71% in 2025.
Global Biosimilars Market Size and Future Outlook
- 2025 Market Size: US$ 34.57 Billion
- 2033 Projected Market Size: US$99.3 Billion
- CAGR (2026–2033): 14.1%
- Dominating Market: North America
- Fastest Growing Market: Asia-Pacific
Market Dynamics
Government-Led Switching and Tender Policies Accelerating Biosimilars Adoption in Public Health Systems
Government procurement and switching policies are playing a major role in accelerating biosimilar uptake, particularly in publicly funded healthcare systems. Many national health authorities now use centralized tendering to purchase biologics in bulk, awarding contracts to lower-priced biosimilars and encouraging hospitals to transition patients from originator drugs. For example, several European public health systems have achieved biosimilar market shares exceeding 70–80% for certain molecules within a few years of tender implementation. These policies enable significant cost savings, allowing healthcare systems to treat larger patient populations within fixed pharmaceutical budgets.
Mandatory or strongly encouraged switching programs are also driving adoption by standardizing prescribing practices in hospitals. In Norway, national tender frameworks enabled biosimilars of infliximab to capture more than 80% of hospital usage within a short period after introduction, demonstrating the impact of coordinated procurement and clinical guidance. Similar public-sector strategies are being expanded in multiple regions, where payers link reimbursement levels or hospital funding incentives to biosimilar prescribing rates. As biologics spending continues to rise globally, such government-led procurement models are expected to remain one of the most influential drivers of biosimilar market growth.
Market Segmentation
The global biosimilars market is segmented based on drug class, therapeutic area, manufacturing modality, route of administration, end-user and region.
Oncology Therapeutic Area Drive Sustained Demand for Biosimilars
The oncology segment is a major driver of biosimilar demand because biologic therapies such as monoclonal antibodies are widely used in cancer treatment and account for a large share of oncology drug expenditure. Globally, more than 20 million new cancer cases are diagnosed each year, creating sustained demand for cost-effective treatment options. Many widely used oncology biologics, including trastuzumab, bevacizumab, and rituximab, have lost patent protection, enabling biosimilar entry and expanding patient access. In several healthcare systems, hospital procurement programs increasingly prioritize biosimilars to manage rising oncology treatment costs while maintaining therapeutic outcomes.
Another important factor is the significant price reduction achieved with biosimilars, which directly improves treatment access. Studies from public health agencies in Europe have shown that biosimilar competition has reduced prices of certain oncology biologics by 30–60%, allowing more patients to receive biologic therapy within fixed healthcare budgets. In the U.S. alone, more than 1.9 million new cancer cases are diagnosed annually, sustaining strong demand for oncology biologics and their biosimilar versions. Increasing inclusion of biosimilars in clinical treatment guidelines and reimbursement frameworks is further accelerating adoption across hospitals and cancer centers.
Geographical Penetration
Largest Market:
Demand for Biosimilars Market in North America
Adoption of biosimilars across North America is accelerating due to payer pressure to reduce biologic drug expenditure and improve patient access to advanced therapies. The U.S. Food and Drug Administration had approved more than 45 biosimilars by 2025, covering major therapeutic classes such as oncology, immunology, and endocrinology. Data from the U.S. Centers for Medicare & Medicaid Services shows that biologics account for over 40% of total Medicare Part B drug spending despite representing a small share of prescriptions, creating strong incentives for biosimilar substitution.
In Canada, public drug plans increasingly prioritize biosimilar switching policies to control healthcare budgets. Provincial programs in British Columbia and Alberta have reported switching rates exceeding 80% for certain biologics following mandatory transition initiatives. These policy actions, combined with increasing physician familiarity, are supporting sustained biosimilar demand across hospitals and specialty clinics.
U.S. Biosimilars Market Outlook
In the U.S., biosimilars are gaining traction in high-cost therapeutic areas, particularly oncology and autoimmune diseases. The FDA reported that biosimilars referencing filgrastim and trastuzumab have achieved significant uptake in hospital oncology settings, supported by clinical equivalence data and payer incentives. According to the U.S. Government Accountability Office, biosimilars generated several billion dollars in savings for Medicare and commercial payers between 2015 and 2023 through price competition and expanded access.
Large health systems and group purchasing organizations are increasingly adopting biosimilars in formularies to reduce procurement costs. In addition, the U.S. Inflation Reduction Act has strengthened pricing scrutiny for high-cost biologics, indirectly encouraging biosimilar adoption as payers seek lower-cost alternatives. Expansion of domestic biologics manufacturing facilities is also improving supply reliability and reducing procurement risks.
Canada Biosimilars Market Trends
Canada’s biosimilar landscape is shaped by coordinated provincial reimbursement reforms and national pharmacare discussions focused on cost containment. The Patented Medicine Prices Review Board has reported that biologic medicines represent roughly one-third of total patented drug sales in Canada, highlighting the financial importance of biosimilar competition. Several provinces, including Ontario and Quebec, have implemented biosimilar transition programs covering insulin glargine, infliximab, and etanercept, significantly increasing biosimilar utilization in public plans.
Health Canada continues to expand approvals for biosimilars, particularly in oncology and rheumatology, improving therapeutic options for patients. Hospital procurement groups are also shifting toward biosimilar-first purchasing policies to manage rising specialty drug expenditures. Growing clinical confidence, coupled with centralized purchasing and reimbursement reforms, is expected to sustain long-term biosimilar demand across Canadian healthcare systems.
Fastest Growing Market:
Asia-Pacific Records the Fastest Growth in the Biosimilars Market
Demand for biosimilars in Asia-Pacific is increasing steadily as healthcare systems aim to reduce biologics expenditure while treating large patient populations. The region accounts for more than 60% of the global population, creating substantial demand for affordable biologic therapies across oncology, autoimmune, and metabolic diseases. Cancer burden remains a major driver, with Asia reporting roughly 9–10 million new cancer cases annually, which significantly increases the need for monoclonal antibody treatments and their biosimilars.
Governments across countries such as Japan, South Korea, China, and India are expanding reimbursement frameworks and accelerating regulatory pathways to improve access to lower-cost biologics. Expansion of regional biologics manufacturing hubs and contract manufacturing capacity is further strengthening supply availability. Increasing physician confidence in biosimilar efficacy and safety is also supporting broader hospital adoption across therapeutic areas.
China Biosimilars Market Insights
China is one of the fastest-growing biosimilar markets, supported by strong regulatory reforms and large domestic demand for biologics. The country reports more than 4.8 million new cancer cases annually, creating sustained demand for oncology biologics and biosimilars in hospital settings. The National Medical Products Administration has approved over 30 biosimilars in recent years, reflecting a rapidly expanding pipeline and improving regulatory efficiency.
Centralized procurement and price negotiations in the national reimbursement system are encouraging hospitals to adopt cost-effective biosimilar alternatives. Domestic biopharmaceutical companies are investing heavily in monoclonal antibody manufacturing facilities, particularly in major biotech clusters such as Shanghai and Jiangsu. These structural factors are enabling biosimilars to play an increasingly important role in China’s long-term strategy to control specialty drug costs and expand treatment access.
India Biosimilars Market Industry Growth
India is emerging as a major biosimilar production and consumption hub due to strong domestic manufacturing capabilities and rising disease burden. The country has approved more than 100 biosimilar products across therapeutic categories, one of the highest numbers globally, reflecting a mature regulatory and development ecosystem. India records over 1.4 million new cancer cases annually, which is driving growing demand for cost-effective oncology biologics in both public and private healthcare facilities.
Government initiatives encouraging domestic biopharmaceutical production and export competitiveness are strengthening investments in biologics manufacturing infrastructure. Indian companies are exporting biosimilars to more than 100 countries, increasing production scale and reducing treatment costs. Expansion of tertiary hospitals, improved insurance coverage, and greater physician familiarity with biosimilars are collectively supporting sustained growth in biosimilar utilization across the country.
Regional Innovation Momentum Index
| Region | Innovation Strength | Pipeline / Innovation Activity | Adoption & Healthcare Impact Indicators |
| North America (U.S.) | FDA approved a record 19 biosimilars in 2024, reflecting accelerating regulatory throughput and policy reforms to simplify interchangeability pathways. | Over 38 biosimilar applications under review globally, with strong participation from companies targeting U.S. approvals. | Biosimilars have generated tens of billions of dollars in healthcare savings in the U.S., supporting payer-driven adoption and formulary expansion. |
| Europe | EMA historically approved 55+ biosimilars over the early development phase of the market, establishing Europe as the most mature regulatory ecosystem. | Strong tender-based procurement systems and early patent expiries continue to stimulate biosimilar launches across oncology and immunology. | Several European health systems report high therapeutic substitution rates, especially in oncology and anti-TNF therapies, reinforcing sustained uptake. |
| Asia-Pacific (China, India, South Korea) | National regulators have issued biosimilar development guidelines and accelerated review pathways to support domestic manufacturing expansion. | Rapid increase in local manufacturers and biologics production capacity, particularly in China and India, with companies exporting to regulated markets. | Lower-cost biosimilars are increasingly used to expand access to biologic therapies in oncology and autoimmune diseases across public healthcare systems. |
Competitive Landscape
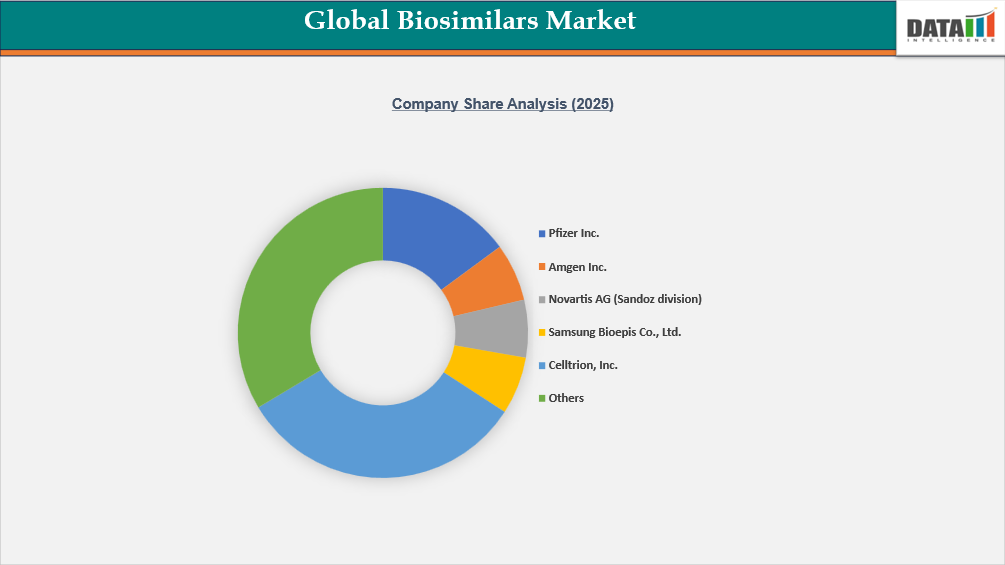
The global biosimilars market is characterized by intense competition among multinational pharmaceutical companies and specialized biosimilar developers, with firms such as Pfizer Inc., Amgen Inc., Novartis AG (Sandoz division), Samsung Bioepis Co., Ltd., and Celltrion, Inc. leading in monoclonal antibody and oncology biosimilars. Companies including Biocon Limited, Viatris Inc., Teva Pharmaceutical Industries Ltd., and Dr. Reddy’s Laboratories Ltd. are strengthening their positions through cost-efficient manufacturing, emerging-market penetration, and strategic partnerships for global commercialization.
European players such as STADA Arzneimittel AG and Fresenius Kabi AG are leveraging strong regional distribution networks and hospital-focused portfolios, while Boehringer Ingelheim International GmbH, Merck KGaA, and F. Hoffmann-La Roche Ltd. maintain influence through biologics expertise, contract manufacturing capabilities, and innovation pipelines. Increasing patent expiries of blockbuster biologics, expanding regulatory pathways, and rising tender-based procurement are intensifying price competition, driving collaborations, and accelerating biosimilar launches across oncology, immunology, and metabolic disease segments.
Investment Landscape
| Company | Year | Development | Impact on Market | |
| Samsung Bioepis Co., Ltd. | 2025 | Expanded biosimilar manufacturing capacity in South Korea to support oncology and immunology pipelines. | Increased global supply capability and strengthened Asia-Pacific export potential. | |
| Biocon Ltd | 2025 | Invested in expanding biologics and biosimilar production facilities in Bengaluru to scale monoclonal antibody output. | Improved cost-competitive supply and supported global tender participation. | |
Key Developments
- In June 2025, Alvotech and Dr. Reddy’s Laboratories entered into a collaboration and license agreement to co-develop, manufacture, and commercialize a biosimilar candidate to pembrolizumab (Keytruda) for global markets. The partnership combines both companies’ biosimilar development and manufacturing capabilities, enabling faster pipeline advancement and broader commercialization reach. With Keytruda generating approximately US$ 29.5 billion in global sales in 2024, such collaborations highlight the strong commercial potential of oncology biosimilars and the growing industry focus on high-value biologic targets.
- In December 2025, Formycon AG and MS Pharma entered an exclusive licensing and supply agreement to commercialize FYB206, a biosimilar candidate to pembrolizumab (Keytruda), across the Middle East and North Africa region. The partnership leverages Formycon’s biosimilar development expertise and MS Pharma’s regional manufacturing and commercialization capabilities to improve access to oncology biologics. As FYB206 approaches completion of clinical development with primary endpoint results expected in early 2026, the agreement reflects the growing trend of regional commercialization partnerships to accelerate biosimilar adoption in emerging markets.
What Sets This Global Biosimilars Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, covering global demand by key segmentation, with region-wise analysis across North America, Europe, Asia-Pacific, South America, and the Middle East & Africa.
- Regulatory Intelligence – In-depth assessment of global pharmaceutical regulatory frameworks impacting market development and commercialization, including FDA, EMA, NMPA, PMDA, and CDSCO requirements, clinical trial pathways, labeling standards, patent exclusivity, and post-marketing surveillance.
- Competitive Benchmarking – Structured benchmarking of leading innovator and generic manufacturers based on product portfolios, pipeline strength, geographic reach, pricing strategies, clinical differentiation, and partnerships in the market.
- Geographic & Emerging Market Coverage – Regional analysis highlighting key impacting factors, with special focus on growth opportunities in Asia-Pacific, Latin America, and Middle Eastern markets.
- Actionable Strategies & Cost Dynamics – Strategic insights into lifecycle management, generic entry risks, combination therapy positioning, pricing pressures, and manufacturing cost structures, supported by expert perspectives from various specialists, regulatory advisors, and pharmaceutical executives.