Global Prurigo Nodularis Treatment Market – Industry Trends & Outlook
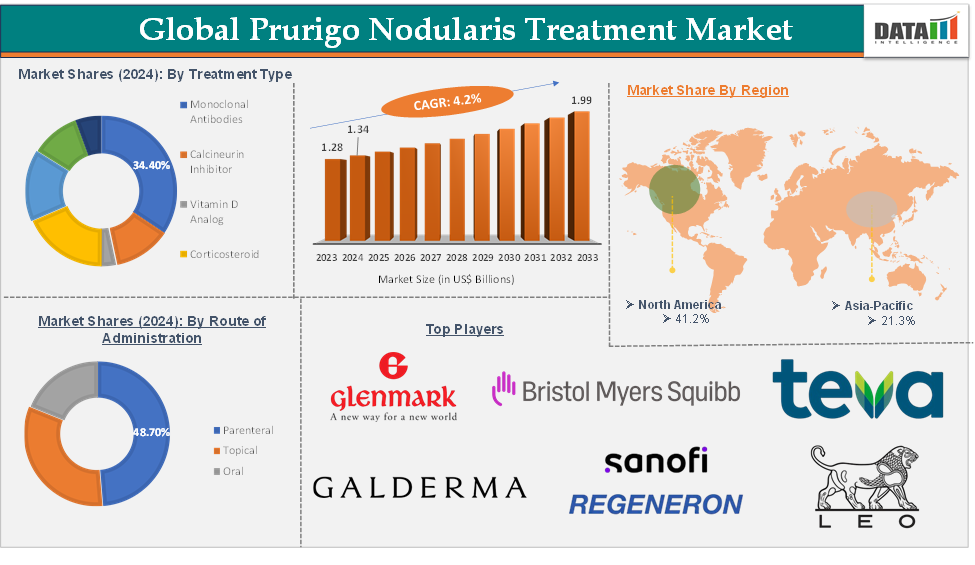
The global prurigo nodularis treatment market was valued at US$ 1.28 Billion in 2023. The market size reached US$ 1.34 Billion in 2024 and is expected to reach US$ 1.99 Billion by 2033, growing at a CAGR of 4.2% during the forecast period 2025-2033.
The global prurigo nodularis treatment market is being propelled by several key factors, including a growing patient population, increased awareness, earlier diagnosis of the disease, and a surge in investment in dermatology research and development. Additionally, the rising incidence of chronic skin diseases due to an aging global population and improved diagnostic capabilities is further fueling market expansion.
A major trend in the prurigo nodularis treatment market is the shift from conventional therapies, such as corticosteroids and immunosuppressants, to advanced biologic and targeted therapies that offer improved efficacy and safety profiles. There is also growing adoption of teledermatology services, expansion of personalized medicine, and increased regulatory approvals for new treatment options. Significant opportunities exist in the development and commercialization of new biologics, monoclonal antibodies, and immune-modulating drugs, as well as in expanding access to advanced therapies in emerging markets where awareness and healthcare infrastructure are improving.
Global Prurigo Nodularis Treatment Market – Executive Summary
Global Prurigo Nodularis Treatment Market Dynamics: Drivers
Rising prevalence of prurigo nodularis
As per JAAD International data in February 2025, Current studies report a wide range of prevalence rates for prurigo nodularis (PN) in adults, varying from 32.7 to 72 cases per 100,000 people. This research seeks to provide a more accurate estimate of the real-world prevalence of PN in the United States, with the goal of confirming whether PN meets the criteria for a rare disease defined as affecting fewer than 200,000 individuals nationwide.
This growth is attributed to factors such as an aging population, higher rates of underlying skin disorders like atopic dermatitis and eczema, and greater awareness and recognition of prurigo nodularis among healthcare professionals and patients. As more people are diagnosed, especially those with moderate-to-severe disease who often do not respond to standard therapies, the demand for effective and innovative treatments continues to rise, fueling market expansion and encouraging pharmaceutical investment in new therapies and advanced biologic agents.
Global Prurigo Nodularis Treatment Market Dynamics: Restraints
High treatment costs
High treatment costs represent a significant restraint for the global prurigo nodularis treatment market. Many of the most effective therapies for prurigo nodularis, such as newly approved biologics and targeted monoclonal antibodies, come with substantial price tags. These advanced treatments often require ongoing administration and specialist supervision, further increasing the overall cost of care.
For patients without comprehensive insurance coverage or in regions with limited healthcare reimbursement, the high out-of-pocket expenses can be prohibitive, leading to underutilization of these therapies. As a result, some patients may continue to rely on less effective, older treatments or may forgo treatment altogether, limiting the market’s growth potential and preventing optimal disease management. Addressing affordability and improving access to innovative therapies are, therefore, critical challenges for stakeholders in this market.
Global Prurigo Nodularis Treatment Market Dynamics: Opportunities
Combination therapies
Combination therapies present a significant opportunity in the prurigo nodularis treatment market because they address the complex and multifactorial nature of the disease, which often involves both immune and neural pathways. Traditional single-agent treatments such as topical corticosteroids, phototherapy, or immunosuppressants frequently provide only partial relief, especially in patients with moderate-to-severe or treatment-resistant prurigo nodularis.
According to the National Center for Biotechnology Information (NCBI), in May 2025, Biologics and JAK inhibitors are transforming the management of prurigo nodularis by providing sustained relief through both immune and neurogenic pathway modulation. For patients with refractory disease, JAK inhibitors offer the advantage of rapid symptom control. Given the diverse nature of prurigo nodularis, these advances highlight the need for personalized treatment strategies tailored to each patient’s unique immunologic profile.
Additionally, novel combinations like antihistamines with leukotriene receptor antagonists (e.g., fexofenadine plus montelukast) have demonstrated benefit in some cases. As new drugs with different mechanisms of action become available, the potential to tailor combination regimens to individual patient needs will expand, offering improved outcomes for those who do not respond to monotherapy and opening new avenues for market growth.
For more details on this report, Request for Sample
Global Prurigo Nodularis Treatment Market – Pipeline Analysis
Global Prurigo Nodularis Treatment Market - Segment Analysis
The global prurigo nodularis treatment market is segmented based on treatment type, route of administration, distribution channel, and region.
Treatment Type:
The monoclonal antibodies treatment type segment is expected to hold 34.3% of the global prurigo nodularis treatment market in 2024
The monoclonal antibodies treatment segment for prurigo nodularis refers to the use of laboratory-engineered antibodies that specifically target key immune or neuroimmune pathways involved in the disease’s pathogenesis. These therapies, such as dupilumab (targeting IL-4/IL-13) and nemolizumab (targeting IL-31), are designed to disrupt the cycle of chronic itch and nodule formation by modulating the immune response and neural signaling associated with prurigo nodularis. This segment also includes investigational agents like vixarelimab and CDX-0159, which act on other relevant pathways.
Monoclonal antibodies have demonstrated significant improvements in itch reduction, lesion clearance, and overall quality of life for patients with refractory or severe disease. Their ability to precisely target the underlying mechanisms of prurigo nodularis, especially type 2 inflammation and neuroimmune dysregulation, has transformed the treatment landscape, offering hope for long-term disease control and better patient outcomes.
For instance, in January 2025, Chime Biologics, a global contract development and manufacturing organization (CDMO), and Mabgeek, an innovative biotech company focused on antibody drug development, completed Process Performance Qualification (PPQ) for MG-K10, a humanized monoclonal antibody targeting IL-4Rα. These factors have solidified the segment's position in the global prurigo nodularis treatment market.
Route of Administration:
The parenteral route of administration segment is expected to hold 48.7% of the global prurigo nodularis treatment market in 2024
The parenteral route of administration segment in the global prurigo nodularis treatment market refers to therapies that are delivered by injection, either subcutaneously (under the skin) or intravenously (into a vein), rather than by oral or topical means. This segment includes biologic drugs such as monoclonal antibodies (e.g., nemolizumab, dupilumab) and other injectable agents designed to target specific immune or neuroimmune pathways involved in prurigo nodularis.
The primary driver for the parenteral route of administration segment is the increasing adoption of advanced biologic therapies that require injection for optimal efficacy and bioavailability. Many patients with moderate-to-severe or refractory prurigo nodularis do not respond adequately to oral or topical treatments, creating a strong demand for injectable options that can deliver targeted, systemic effects.
The proven effectiveness of parenteral biologics in reducing itch, healing nodules, and improving quality of life is fueling growth in this segment, as both healthcare providers and patients seek therapies that offer durable and meaningful symptom relief.
Global Prurigo Nodularis Treatment Market – Geographical Analysis
North America is expected to hold 41.2% of the global prurigo nodularis treatment market in 2024
Prurigo nodularis is a chronic, underdiagnosed neuroimmune skin disorder estimated to affect up to 170,800 people in the United States. The North American prurigo nodularis treatment market is experiencing strong growth, making it the largest regional market globally. Key drivers include the rising prevalence of prurigo nodularis, particularly as the population ages and awareness of the disease increases among both healthcare professionals and patients.
Improved diagnostic procedures and educational initiatives have led to higher diagnosis rates and better identification of patients needing treatment. Another major driver is the rapid advancement and adoption of innovative therapies, especially biologics and monoclonal antibodies like nemolizumab and dupilumab, which offer improved efficacy and safety compared to traditional treatments. Substantial investment in research and development, a robust clinical pipeline, and frequent regulatory approvals are further accelerating market expansion.
Additionally, rising healthcare expenditures in North America, particularly in the United States, support broader access to advanced treatments and drive demand for new therapeutic options. The increasing use of telemedicine and digital health platforms also enhances patient access to specialized dermatological care, contributing to overall market growth in the region.
For instance, in August 2024, the U.S. Food and Drug Administration (FDA) approved Galderma's Nemluvio (nemolizumab) as a pre-filled pen for subcutaneous injection for the treatment of adults with prurigo nodularis. Thus, the above factors are consolidating the region's position as a dominant force in the global prurigo nodularis treatment market.
Asia Pacific is expected to hold 21.3% of the global prurigo nodularis treatment market in 2024
The Asia-Pacific prurigo nodularis treatment market is experiencing rapid growth, driven by several key factors. Rising disease awareness among both healthcare professionals and the general population has led to earlier diagnosis and increased demand for effective therapies.
Expanding healthcare infrastructure and greater government investment in chronic disease management are improving patient access to specialized dermatological care and advanced treatments. The region’s large and growing patient population, particularly in populous countries like China and India, creates significant market potential, especially as the incidence of prurigo nodularis is recognized to be substantial in these areas.
Another major driver is the introduction and adoption of innovative targeted therapies, such as biologics like nemolizumab and dupilumab, which address the underlying immunological mechanisms of the disease rather than just providing symptomatic relief.
Pharmaceutical companies are increasing investments in research, clinical trials, and collaborations to expand their product pipelines and bring new therapies to market. Digital health initiatives, teledermatology, and educational campaigns by organizations such as the International Forum for the Study of Itch (IFSI) and the Asian Academy of Dermatology and Venereology (AADV) are further supporting disease recognition and treatment uptake.
For instance, in June 2024, Maruho Co., Ltd. announced the launch of Mitchga Subcutaneous Injection 30mg Vials in Japan. Mitchga contains nemolizumab, a humanized monoclonal antibody that targets the interleukin-31 (IL-31) receptor A. This product is indicated for the treatment of pruritus (itching) associated with atopic dermatitis in pediatric patients (ages 6 to under 13) and for prurigo nodularis in adults and children aged 13 and older, specifically when existing treatments are insufficiently effective.
Global Prurigo Nodularis Treatment Market – Competitive Landscape (Major Players)
The major global players in the prurigo nodularis treatment market include Sanofi and Regeneron Pharmaceuticals, Inc., Galderma, Teva Pharmaceuticals USA, Inc., Glenmark Pharmaceuticals Inc., USA, LEO Pharma, GSK plc., Abbott, and Bristol Myers Squibb company. Among others.
Global Prurigo Nodularis Treatment Market – Competitive Landscape (Emerging Players)
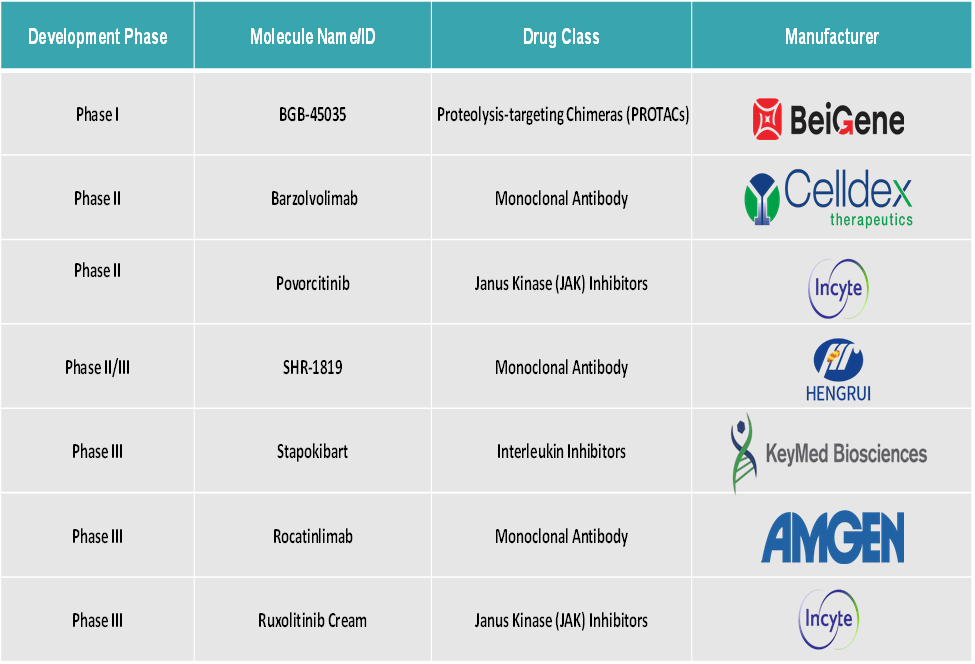
The emerging players in the prurigo nodularis treatment market include BeiGene, Celldex Therapeutics, Incyte Corporation, Guangdong Hengrui Pharmaceutical Co., Ltd, Keymed Biosciences Co., Ltd, and Amgen, among others.
Global Prurigo Nodularis Treatment Market – Key Developments
In February 2025, the Medicines and Healthcare products Regulatory Agency (MHRA), the UK’s medicines regulator, granted marketing authorisation for nemolizumab (brand name Nemluvio) for two major skin conditions: moderate-to-severe prurigo nodularis in adults and moderate-to-severe atopic dermatitis (eczema) in both adolescents (aged 12 and older, weighing at least 30 kg) and adults.
In September 2024, Amber Specialty Pharmacy announced the availability of Galderma's latest medication, NEMLUVIO (nemolizumab-ilto), for the treatment of Prurigo Nodularis in adults. This partnership marks a significant advancement in the therapeutic options available to patients suffering from this chronic, debilitating skin condition.
In March 2024, Incyte announced new clinical results for its investigational drug, povorcitinib (INCB54707), an oral JAK1 inhibitor. The results come from a Phase 2 clinical trial assessing the drug’s effectiveness and safety in adult patients with prurigo nodularis (PN).
In February 2024, Galderma announced that the U.S. Food and Drug Administration (FDA) had accepted its Biologics License Applications for nemolizumab, intended for the treatment of prurigo nodularis as well as moderate to severe atopic dermatitis in both adolescents and adults. Nemolizumab is a first-in-class investigational monoclonal antibody engineered to block IL-31 signaling, offering safe and rapid relief from the most troublesome symptom associated with both conditions: persistent itch.
Global Prurigo Nodularis Treatment Market – Scope
Metrics | Details | |
CAGR | 4.2% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Bn) | |
Segments Covered | Treatment Type | Monoclonal Antibodies, Calcineurin Inhibitor, Vitamin D Analog, Corticosteroid, Immunomodulatory Drugs (IMiDs), Antihistamines, Others |
Route of Administration | Parenteral, Topical, Oral | |
Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
DMI- Insights
The global prurigo nodularis treatment market is witnessing significant growth, with market valuations estimated at US$ 1.34 billion in 2024 and expected to reach US$ 1.40 billion by 2025, with projections soaring to US$ 1.99 billion by 2033. This expansion is fueled by increasing disease prevalence, heightened awareness among both healthcare professionals and patients, and the launch of innovative therapies, notably biologics and targeted monoclonal antibodies. North America leads the market, to its sizable aging population, rapid uptake of advanced treatments, and robust investment in dermatological research.
Emerging trends in the market include a strong movement toward personalized medicine, a rise in regulatory approvals for next-generation therapies, and the growing use of teledermatology to enhance patient access. Pharmaceutical companies are ramping up research and development, resulting in a dynamic pipeline of new treatments with better efficacy and safety.
Despite these advancements, the market continues to face obstacles such as high therapy costs, reimbursement limitations in certain regions, and insufficient physician training, all of which hinder broader access, especially in developing countries.
The global prurigo nodularis treatment market report delivers a detailed analysis with 56 key tables, more than 54 visually impactful figures, and 173 pages of expert insights, providing a complete view of the market landscape.
Suggestions for Related Report
For more pharmaceutical-related reports, please click here