Market Size
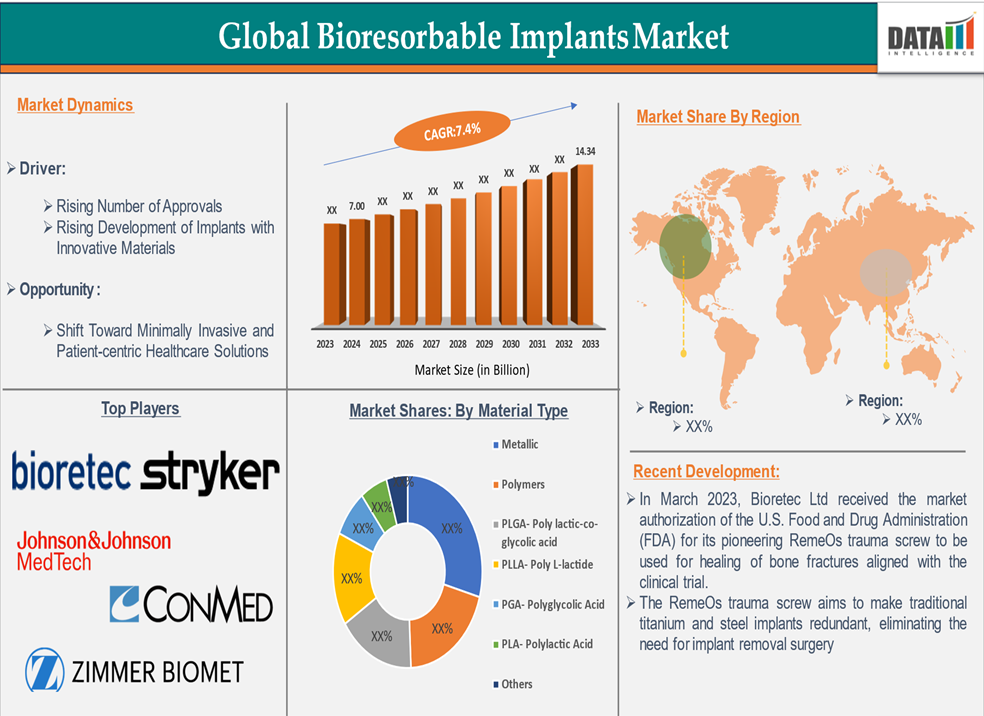
The Bioresorbable Implants Market size reached US$ 7.00 billion in 2024 and is expected to reach US$ 14.34 billion by 2033, growing at a CAGR of 7.4% during the forecast period 2025-2033.
Bioresorbable implants are medical devices made from specially engineered polymers that naturally dissolve or degrade within the body over time, eliminating the need for surgical removal once their function is complete. These implants are composed of either hydrophobic or hydrophilic polymers and are designed to support healing processes in various medical applications, particularly after surgery.
They are available in a wide range of forms and are commonly used in orthopedic procedures, tissue repair, and drug delivery systems. As they gradually break down into non-toxic byproducts that are safely absorbed or excreted by the body, bioresorbable implants offer a less invasive, patient-friendly alternative to permanent implants, reducing the risk of long-term complications and the need for additional surgeries.
Executive Summary
For more details on this report – Request for Sample
Market Dynamics: Drivers & Restraints
Rising Development of Advanced Implants with Innovative Materials
The rising development of implants utilizing innovative materials is significantly driving the growth of the bioresorbable implants market. Advancements in material science have led to the creation of bioresorbable polymers and composites that offer improved mechanical strength, biocompatibility, and controlled degradation rates. For instance, the integration of bioactive glass and magnesium-based alloys into implant designs has enhanced their functionality and healing capabilities.
These innovations enable the development of implants that not only support healing but also promote tissue regeneration, thereby expanding their applications across various medical specialties. In January 2025, Bioretec, a pioneer in absorbable orthopedic implants, has completed its CE mark approval process and can start commercialization of its RemeOs Trauma Screw product portfolio within the European Union and non-European countries that recognize the CE mark market authorization. This comprehensive approval covers all cannulated and non-cannulated product designs, with sizes ranging from diameters of 2.0mm to 4.0mm and lengths from 8mm to 50mm. Indications approved include the use of these screws for fracture and malalignment fixations in both upper and lower extremities of adult and pediatric patients, excluding the hand and forefoot.
Additionally, the incorporation of smart technologies, such as sensors and drug delivery systems, into bioresorbable implants allows for real-time monitoring and targeted treatment, further enhancing patient outcomes. These technological advancements are making bioresorbable implants more effective and versatile, thereby accelerating their adoption in clinical settings.
High Cost of Designing and Manufacturing
The high cost of designing and manufacturing bioresorbable implants remains a major barrier to the market's broader adoption. These implants require advanced materials, specialized production techniques, and extensive research and testing to ensure biocompatibility and precise degradation rates. Processes like 3D printing, polymer blending, and surface modification demand sophisticated equipment and skilled labor, all of which significantly drive up production costs.
Market Segment Analysis
The Global Bioresorbable Implants Market is segmented based on material type, application, and end-user region.
By Material Type:
PLGA (Poly lactic-co-glycolic acid) in the material type segment is expected to dominate the bioresorbable implants market.
PLGA (Poly lactic-co-glycolic acid) is expected to dominate the bioresorbable implants market due to its exceptional versatility, safety profile, and broad range of clinical applications. As a copolymer of lactic acid and glycolic acid, PLGA offers customizable degradation rates by simply altering the ratio of its two components, allowing it to be tailored for everything from short-term tissue scaffolds to longer-term orthopedic or cardiovascular support. Its biocompatibility and non-toxic degradation byproducts (carbon dioxide and water) make it ideal for implantation, with minimal risk of inflammation or rejection.
PLGA is already widely used in FDA-approved medical devices, including sutures, screws, pins, and drug-delivery systems, which has established a strong regulatory precedent for new product approvals. Moreover, PLGA’s role in controlled drug release has expanded its utility beyond just structural support. Given its proven track record, ease of processing, and adaptability across various specialties like orthopedics, cardiology, and regenerative medicine, the PLGA segment is well-positioned to remain a leading force in the global bioresorbable implants market.
Market Geographical Share
North America is expected to dominate the global bioresorbable implants market.
North America is expected to dominate the bioresorbable implants market due to rising surgical volumes, growing adoption of advanced medical technologies, and increasing demand for patient-friendly treatment options. The region, particularly the United States, has seen a steady rise in procedures such as orthopedic repairs, soft tissue reconstruction, and cardiovascular interventions.
For instance, according to the American College of Rheumatology, in 2024, about 790,000 total knee replacements and 544,000 hip replacements were done every year in the U.S. This number continues to grow as our population ages. This increase in the number of procedures is expected to increase the demand for bioresorbable implants, as a large number of individuals in the region are hesitating about the removal surgery of implants.
Additionally, a strong focus on reducing long-term complications and avoiding secondary surgeries has led to higher acceptance of bioresorbable devices like screws, pins, and plates. Recent product approvals, such as magnesium-based bioresorbable implants, and growing use of materials like PLGA and PLLA further support this trend.
With a well-established regulatory pathway and a high level of awareness among healthcare providers, North America is expected to maintain its leading position in the global bioresorbable implants market. For instance, In March 2023, Bioretec Ltd received the market authorization of the U.S. Food and Drug Administration (FDA) for its pioneering RemeOs trauma screw to be used for healing of bone fractures aligned with the clinical trial.
Market Companies
The global market players in the bioresorbable implants market include Bioretec Ltd, Stryker Corporation, CONMED Corporation, Syntellix AG, Johnson & Johnson Medical Devices (DePuy Synthes), Zimmer Biomet, NuVasive Inc., KLS Martin Group, Smith+Nephew, Evonik Industries AG, among others.
Recent Developments:
April 2026: Biodegradable implant coatings and materials are gaining strong traction, with growing adoption of succinic-based bioresorbable coatings to enhance implant performance, biocompatibility, and controlled degradation rates in orthopedic and cardiovascular applications.
March 2026: Bioretec Ltd announced continued expansion of its bioresorbable orthopedic implant portfolio, supported by regulatory clearances and commercialization of its PLGA-based Activa implants for broader trauma and pediatric applications.
March 2026: Investment activity increased in the sector, with funding initiatives aimed at accelerating biodegradable orthopedic implant innovation, particularly focusing on next-generation materials that eliminate secondary removal surgeries and improve healing outcomes.
February 2026: Advancements in bioresorbable polymer technologies are driving new product development, with rising clinical adoption of polymer-based implants (such as stents, screws, and scaffolds) due to improved safety profiles and minimally invasive treatment benefits.
Market Scope
| Metrics | Details | |
| CAGR | 7.4% | |
| Market Size Available for Years | 2024-2033 | |
| Estimation Forecast Period | 2026-2033 | |
| Revenue Units | Value (US$ Bn) | |
| Material Type | Metallic, Polymers, PLGA- Poly lactic-co-glycolic acid, PLLA- Poly L-lactide, PGA- Polyglycolic Acid, PLA- Polylactic Acid, Others | |
| Segments Covered | Application | Cardiovascular, Orthopedics, Others |
| End-User | Hospitals, Ambulatory Surgical Centers, Others | |
| Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
Why Purchase the Report?
- Pipeline & Innovations: Reviews ongoing clinical trials, product pipelines, and forecasts upcoming pharmaceutical advancements.
- Type Performance & Market Positioning: Analyzes product performance, market positioning, and growth potential to optimize strategies.
- Real-World Evidence: Integrates patient feedback and data into product development for improved outcomes.
- Physician Preferences & Health System Impact: Examines healthcare provider behaviors and the impact of health system mergers on adoption strategies.
- Market Updates & Industry Changes: Covers recent regulatory changes, new policies, and emerging technologies.
- Competitive Strategies: Analyzes competitor strategies, market share, and emerging players.
- Pricing & Market Access: Reviews pricing models, reimbursement trends, and market access strategies.
- Market Entry & Expansion: Identifies optimal strategies for entering new markets and partnerships.
- Regional Growth & Investment: Highlights high-growth regions and investment opportunities.
- Supply Chain Optimization: Assesses supply chain risks and distribution strategies for efficient Type delivery.
- Sustainability & Regulatory Impact: Focuses on eco-friendly practices and evolving regulations in healthcare.
- Post-market Surveillance: Uses post-market data to enhance product safety and access.
- Pharmacoeconomics & Value-Based Pricing: Analyzes the shift to value-based pricing and data-driven decision-making in R&D.
The global bioresorbable implants market report will provide approximately 45 tables, 46 figures, and 180 pages.
Target Audience
- Manufacturers: Pharmaceutical, Biotech Companies, Contract Manufacturers, Distributors, Hospitals.
- Regulatory & Policy: Compliance Officers, Government, Health Economists, Market Access Specialists.
- Technology & Innovation: R&D Professionals, Clinical Trial Managers, Pharmacovigilance Experts.
- Investors: Healthcare Investors, Venture Fund Investors, Pharma Marketing & Sales.
- Consulting & Advisory: Healthcare Consultants, Industry Associations, Analysts.
- Supply Chain: Distribution and Supply Chain Managers.
- Consumers & Advocacy: Patients, Advocacy Groups, Insurance Companies.
- Academic & Research: Academic Institutions.