At-Home Testing Kits Market Size
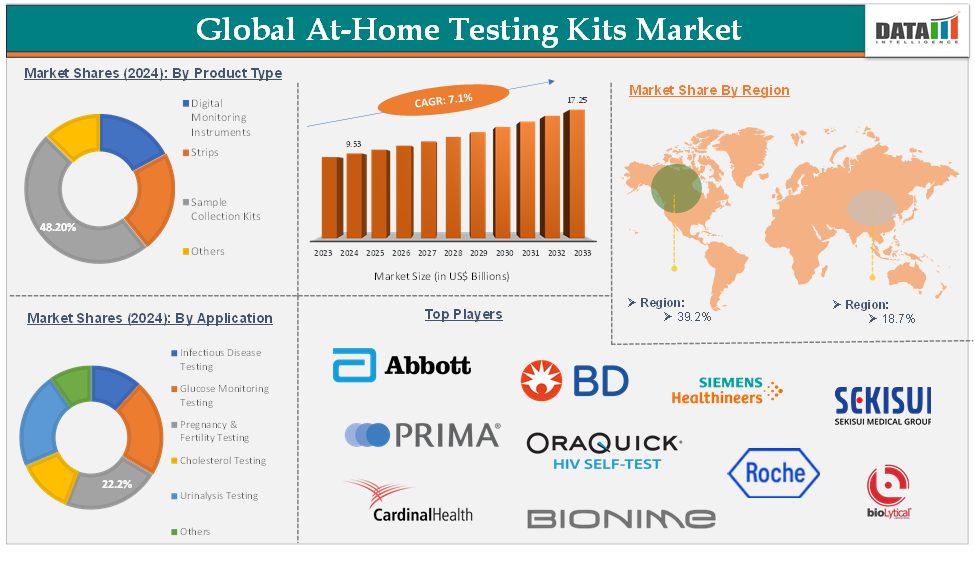
The global At home Testing Kits market was US$ 9.53 Billion in 2025 and expected to reach up to US$ 17.25 Billion in 2033 growing at a CAGR of 7.1% during the forecasting period (2026-2033).
At-home testing kits, also known as home-use tests, are commercially available medical devices that enable individuals to perform health screenings or monitor various conditions from the comfort of their own homes. These kits, which can be purchased online or at pharmacies, empower users to conveniently test for, detect, or track specific diseases or health parameters without the need for a clinical visit.
Technological advancements are a key driver propelling the growth of the global at-home testing kits market over the forecast period. For instance, in February 2024, Masimo’s MightySat Medical recently received FDA clearance, making it the first and only FDA-cleared fingertip pulse oximeter available without a prescription.
This innovation allows users access to a medical-grade pulse oximeter powered by Masimo SET pulse oximetry technology directly at home. Such advancements are making at-home testing procedures simpler, more efficient, and highly accessible. As a result, consumer adoption of these testing kits is rising, further fueling demand and driving the expansion of the global at-home testing kits market.
Chronic conditions such as cardiovascular diseases, cancer, and diabetes remain leading causes of mortality and disability, particularly in developed markets like the United States, where they also drive a significant portion of the nation’s US$4.9 trillion annual healthcare expenditure.
Moreover, 73% of NCD-related deaths occur in low- and middle-income countries, where access to healthcare infrastructure is limited, creating a strong use case for decentralized and affordable at-home testing solutions. Premature deaths linked to NCDs further emphasize the role of early detection and regular monitoring enabled by home diagnostics.
Therefore, the global at-home testing kits market is transitioning from a pandemic-driven necessity to a long-term component of preventive and decentralized healthcare delivery. Rising health awareness, aging populations, and the growing burden of chronic and noncommunicable diseases continue to underpin sustained demand for home-based diagnostics.
Beyond these core drivers, expanding telemedicine adoption is further strengthening the relevance of at-home tests by enabling remote diagnosis and clinical decision-making.
Technological advancements such as digital readouts, smartphone integration, and AI-enabled result interpretation are improving test accuracy and user confidence. Increasing healthcare cost pressures are also encouraging payers and providers to shift diagnostics from hospitals to home settings. Growth is additionally supported by rising penetration in rural and remote regions where access to diagnostic infrastructure remains limited.
Executive Summary
Sources: DataM Intelligence
For more details on this report, Request for Sample
Market Dynamics: Drivers
Expansion of Telehealth and Remote Patient Monitoring
The rapid expansion of telehealth and remote patient monitoring is significantly driving demand for at-home testing kits by reshaping how healthcare is accessed and delivered.
The global telehealth and telemedicine market is projected to grow from US$94.14 billion in 2024 to US$180.86 billion by 2030, reflecting sustained digital healthcare adoption beyond the pandemic. Telehealth usage has structurally shifted, increasing from around 1% of patient visits in early 2020 to nearly 17% by 2023, indicating lasting behavioral change among patients and providers.
As virtual consultations become routine, clinicians increasingly rely on at-home diagnostic data to support remote clinical decision-making. At-home testing kits enable timely disease detection, monitoring, and follow-up without physical hospital visits. This model is particularly effective for chronic disease management, infectious disease screening, and preventive care.
Advances in remote monitoring technologies, including smartwatches and fitness trackers, are further strengthening this ecosystem. These wearables allow continuous sharing of vital health data such as glucose levels, heart rate, oxygen saturation, and activity metrics. Integration of wearable data with telemedicine platforms enhances diagnostic accuracy and personalized care planning.
At-home testing kits complement this by providing episodic clinical validation alongside continuous monitoring. Looking ahead, deeper integration between telehealth platforms, wearable devices, and home diagnostics is expected to accelerate adoption. Improved interoperability, AI-driven analytics, and value-based care models will further boost demand. Thus, above factors helps to boost the market growth.
Advancements in diagnostic technology
Technological advancements are playing a pivotal role in driving the growth of the global at-home testing kits market. As the healthcare sector evolves, there is a rising demand for diagnostic solutions that are rapid, accurate, and easily accessible outside of traditional clinical settings.
Innovations such as biosensors, microfluidics, and lab-on-a-chip systems have significantly enhanced the efficiency, reliability, and user-friendliness of at-home diagnostics. For instance, in September 2024, Abbott Laboratories introduced Lingo, an over-the-counter continuous glucose monitor in the United States. Lingo leverages advanced biosensor technology to provide real-time insights and alert users about sudden changes in blood glucose levels, empowering individuals to manage their health proactively from home.
This type of digital monitoring instrument exemplifies the shift toward smarter, more connected at-home testing solutions. These ongoing advancements are simplifying testing procedures, making them more efficient and accessible for a wider audience. As a result, more people are adopting at-home testing kits, which is accelerating market growth and transforming the way individuals engage with their health.
Market Dynamics: Restraints
Stringent regulatory requirements
Stringent regulatory requirements are a significant barrier to the growth of the global at-home testing kits market. As these kits are classified as medical devices, they must undergo thorough approval processes by regulatory agencies such as the FDA in the United States and their counterparts worldwide. This process typically involves extensive clinical validation to ensure the accuracy and reliability of test results.
However, regulatory frameworks vary considerably across different regions, leading to a fragmented market landscape. Companies aiming for simultaneous market entry in multiple countries face the added complexity of navigating diverse regulatory standards, documentation protocols, and clinical trial requirements unique to each jurisdiction. These challenges collectively slow down product approvals and market expansion, thereby hampering the growth of the global at-home testing kits market.
Market - Segment Analysis
The global at-home testing kits market is segmented based on product type, application, sample, distribution channel, and region.
Product Type:
The sample collection kits product type segment is expected to hold 48.2% of the global at-home testing kits market
Sample collection kits represent a vital segment that facilitates accurate, user-friendly, and hygienic sample gathering for diagnostic analysis. These kits are specifically designed to enable individuals to collect biological samples, such as saliva, blood (via finger prick), urine, or stool, in the comfort of their own homes, without the need for professional assistance.
The effectiveness of at-home diagnostic tests largely depends on the quality and integrity of the collected sample, making the design and reliability of these kits critical. Most kits include essential components such as collection tools (e.g., swabs, lancets, collection tubes), clear instructions, and secure packaging for shipping samples to certified laboratories.
With increasing demand for convenient and non-invasive testing options, especially for chronic conditions, infectious diseases, and wellness monitoring, the sample collection kit segment is experiencing robust growth. Innovations in this space, such as pre-labeled vials, integrated barcoding for digital tracking, and IoT-enabled collection devices, are further enhancing usability and accuracy, positioning this segment as a cornerstone of the expanding digital health ecosystem.
Market Geographical Analysis
North America is expected to hold 39.2% of the global at-home testing kits market
North America is expected to hold a substantial share of the global at-home testing kits market, driven by its robust healthcare infrastructure, high prevalence of chronic diseases, and rapid technological advancements. The growing incidence of conditions such as diabetes, cardiovascular diseases, and infectious illnesses is a key factor fuelling market growth in the region.
According to the Centers for Disease Control and Prevention, as of 2024, over 38 million Americans, approximately 1 in 10, have diabetes, with 90% to 95% diagnosed with type 2 diabetes. Although type 2 diabetes primarily affects adults over 45, it's increasingly seen in children, teens, and young adults, intensifying the demand for accessible and efficient diagnostic solutions like at-home testing kits.
The presence of major industry players further contributes to regional growth through ongoing innovation and product launches. For instance, in June 2024, Prevounce Health introduced the Pylo GL1-LTE, its first remote blood glucose monitoring device. Designed for reliability, the GL1-LTE supports widespread cellular connectivity across the U.S. and integrates seamlessly with the Prevounce remote care management platform.
It also connects with other health software via the Pylo cloud API. Prevounce's product suite, including cellular-enabled blood pressure monitors and weight scales, reflects a broader trend toward user-friendly, tech-enabled diagnostics. These innovations are streamlining testing processes and accelerating the adoption of at-home testing kits throughout North America. Thus, the above factors are consolidating the region's position as a dominant force in the global at-home testing kits market.
Asia-Pacific is expected to hold 18.7% of the global at-home testing kits market
The Asia Pacific region is emerging as the fastest-growing market for at-home testing kits, owing to rising healthcare funds, increased awareness of early disease diagnosis, the demand for quick and precise diagnostic results, and technological advancements. The need for at-home testing kits has increased as these kits have proven invaluable for the early diagnosis and monitoring of a variety of health disorders, thereby improving disease management results throughout the region.
There are recent launches of devices in this region, making it the fastest-growing region. For instance, in September 2024, Mankind Pharma Ltd. announced the launch of its latest innovation, RAPID NEWS self-test kits, which are intended to address common health conditions such as dengue, urinary tract infections (UTIs), and early menopause.
This launch is a significant step toward accessible healthcare, to provide citizens across India with convenient, confidential, and rapid testing alternatives. Thus, the above factors are consolidating the region's position as the fastest-growing force in the global at-home testing kits market.
Market Major Players
The major global players in the at-home testing kits market include Abbott, F. Hoffmann-La Roche Ltd, BD, Siemens Healthcare Private Limited, Geratherm Medical AG, OraQuick, SEKISUI Diagnostics, BioLytical Laboratories Inc. (INSTI), PRIMA Lab SA, Nova Biomedical, Everlywell, Cardinal Health, and Bionime Corporation, among others.
Key Developments
February 2026: Rising adoption of self-diagnostic healthcare solutions and digital health monitoring accelerated growth in the global At-Home Testing Kits market. Consumers increasingly utilized home-based diagnostic kits for infectious diseases, chronic condition monitoring, fertility tracking, and wellness assessment.
January 2026: Increasing investments in rapid diagnostics, AI-enabled interpretation tools, and connected healthcare platforms strengthened innovation in the At-Home Testing Kits market worldwide. Companies expanded development of smartphone-integrated testing kits and app-based result management systems to improve accessibility and convenience.
December 2025: Growing demand for preventive healthcare, personalized medicine, and decentralized diagnostic services accelerated commercialization of advanced at-home testing technologies globally. Healthcare providers increasingly supported home-based testing solutions to reduce hospital visits and improve early disease detection.
November 2025: Expansion of telehealth services and remote patient management programs across North America, Europe, and Asia-Pacific strengthened deployment of at-home diagnostic solutions. Governments and healthcare organizations increasingly promoted home testing initiatives to improve healthcare accessibility in rural and underserved populations.
October 2025: Rising advancements in biosensor technologies, microfluidics, and AI-powered diagnostic analytics improved accuracy and efficiency in the At-Home Testing Kits market globally. Research institutions accelerated development of multiplex testing platforms capable of detecting multiple biomarkers through compact and user-friendly devices.
September 2025: Increasing collaborations between diagnostic companies, digital health providers, and retail pharmacy chains accelerated innovation in at-home testing solutions worldwide. Strategic partnerships focused on connected diagnostics, subscription-based wellness testing, and integrated telemedicine support services.
August 2025: Growing prevalence of chronic diseases and rising consumer awareness regarding proactive health monitoring strengthened expansion of the At-Home Testing Kits market globally. Consumers increasingly adopted home testing kits for diabetes management, cardiovascular risk assessment, hormone analysis, and infectious disease screening.
July 2025: Rising demand for fast, convenient, and privacy-focused healthcare diagnostics accelerated innovation in portable and easy-to-use testing platforms worldwide. Companies expanded development of saliva-based, blood-based, and urine-based diagnostic kits supporting rapid and accurate home testing applications.
June 2025: Increasing focus on preventive healthcare, patient convenience, and decentralized diagnostic infrastructure supported strong growth in the global At-Home Testing Kits market. North America maintained market leadership due to strong consumer awareness and advanced digital healthcare infrastructure, while Asia-Pacific emerged as the fastest growing region driven by expanding healthcare access, rising disposable incomes, and increasing adoption of digital health technologies.
Market Scope
Metrics | Details | |
CAGR | XX% | |
Market Size Available for Years | 2025-2033 | |
Estimation Forecast Period | 2026-2033 | |
Revenue Units | Value (US$ Bn) | |
Segments Covered | Product Type | Digital Monitoring Instruments, Strips, Sample Collection Kits, Others |
Application | Infectious Disease Testing, Glucose Monitoring Testing, Pregnancy & Fertility Testing, Cholesterol Testing, Urinalysis Testing, Others | |
Sample Type | Urine, Blood, Saliva, Vaginal Swab, Others | |
Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |
























































