Interspinous Spacers Market
DMI segments the Interspinous Spacers Market by geography into – North America, South America, Europe, Asia-Pacific (APAC), and Middle East and Africa.
2019-11-21
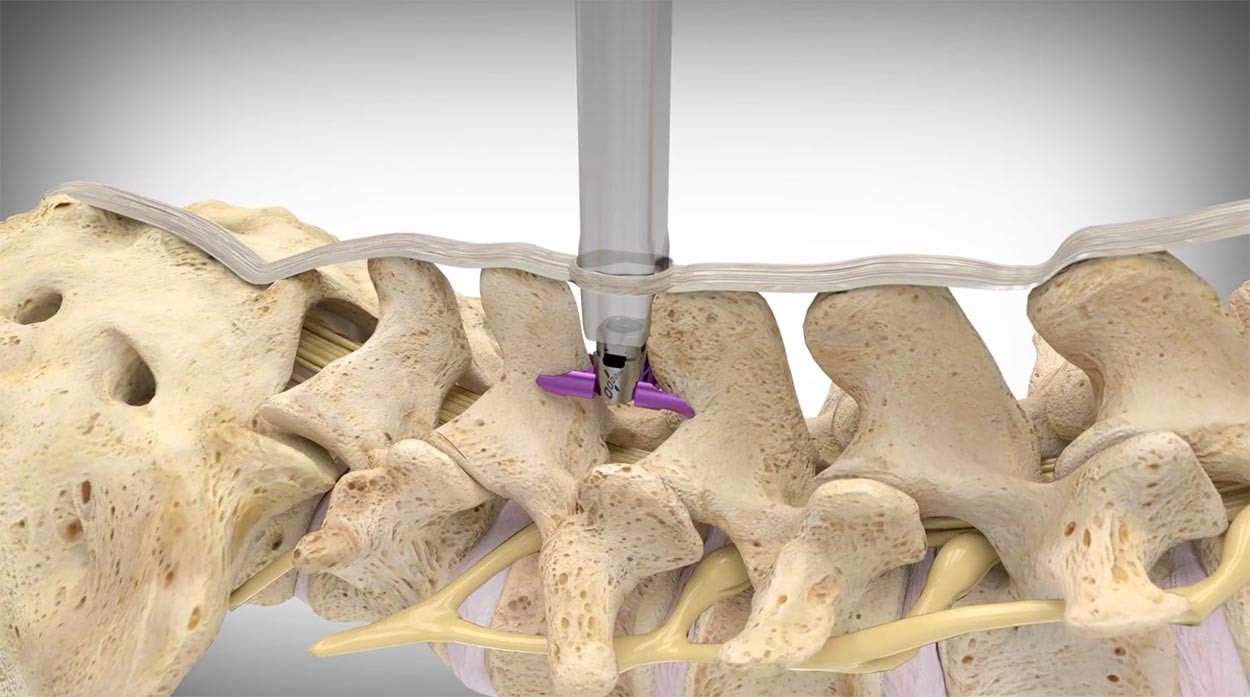
A minimally invasive implant for the treatment of lumbar spinal stenosis
Interspinous spacers are small devices implanted between the vertebral spinous processes. These devices spread the vertebrae apart and keep the space for the nerves open and functioning. By enlarging the space between the bones in the back, the spacers reduce pressure on the spinal nerves, thereby alleviating the symptoms of spinal stenosis such as leg pain and numbness in lower extremities.
Global Interspinous Spacers Market Drivers, Restraints and Market Opportunity
Solely based on Epidemiology and new technological advancements, overcome the setbacks
The market for interspinous spacers is expected to be driven by the increase in the prevalence of degenerative disk disease and spinal stenosis, rise in the aged population along with increasing awareness regarding the need for treatment of spinal disorders.
Lumbar Spinal Stenosis is the most common reason for spinal surgery for patients over 65 years of age with the prevalence of acquired lumbar spinal stenosis in North America being estimated to be almost 20% for the population aged between 60-69 years. The disorder is especially prevalent among the aged population with prevalence rates rising with age and increasing sharply over 60 years of age.
The Non-Invasive nature of the procedure also enables lower surgical procedure and patient recuperation time lead which enables treatment among patients at high risk of surgical complications such as the aged population.
The approval of Medicaid reimbursement for interspinous spacers is also expected to drive the adoption of these devices.
Significant rates of complications associated with the usage of interspinous spacers have resulted in extremely slow regulatory approvals for new interspinous spacers in the North American market. Multiple clinical studies have also shown no significant long-term benefits of using these devices while reporting higher surgical costs and post-surgical complication rates. The US market currently has only 1 FDA approved spacer device (The Superion® InterSpinous Spacer by Boston Scientific Corporation) with most of the device applicants such as Medtronic and Globus Medical Inc. being rejected during clinical trials. A large number of complications have also forced manufacturers to withdraw their products; for instance, Medtronic had to withdraw their X-STOP® Interspinous Process Decompression (IPD) System in 2015 after multiple reports of complications such as spinous process fractures. The market is also facing reimbursement problems with the majority of private insurance providers classifying the procedure as investigational or experimental.
Global Interspinous Spacers Market Segmentation
The global market for Interspinous Spacers is broadly classified into devices type, materials used, indication and end-user.
Based on the device type the market for Interspinous Spacers is classified into Static Interspinous Spacers and Dynamic Interspinous Spacers. Based on materials, Interspinous Spacers is classified into Titanium Spacers, PEEK Spacers, Silicone Spacers, and Others
Lumbar Spinal Stenosis is the major market shareholder amongst all the Interspinous Spacers indications, followed by Degenerative Disk Disease and others. By End-Users the market is classified into Hospitals, Ambulatory Surgical Clinics, Speciality Clinics, and others.
Global Interspinous Spacers Market – Geographical Analysis
With an increase in FDA approvals for innovative devices the patients hoping for the best and most useful technologies
DataM Intelligence regions segmentation covers major regions such as North America, Europe, Asia Pacific, South Africa, and the Middle East and Africa.
North America is the largest market for Interspinous Spacers due to the widespread availability of advanced medical treatments in the region combined with a large target population base and availability of healthcare expense reimbursement. The market is witnessing a rapid rise in the aged population which expected to drive demand for spinal stenosis and DDD treatments.
Asia-Pacific is expected to be the fastest-growing market for interspinous spacers due to the large size of the target patient population combined with increasing investment in healthcare in the region. The market has the largest number of people affected by Degenerative disc diseases and spinal stenosis which is expected to grow further as the population ages. The rapid increase in the aged population is expected to drive demand for interspinous spacers in the future.
Global Interspinous Spacers Market – Competitive Analysis
May o9, 2019: Boston Scientific grabs the spine-spacer developer Vertiflex in $465M deal
List of major market players – Boston Scientific Corporation, RTI Surgical Holdings Inc., Medtronic Plc., Zimmer Biomet Holdings Inc., Orthofix Medical Inc., Globus Medical Inc., Alphatec Spine Inc.
About: DataM Intelligence
DataM Intelligence was established on an elemental idea of publishing Advanced Market Research Reports with precise data points & utmost accuracy that will accelerate the decision making in designing disruptive solutions. This aids us in providing the detailed scenario of the present and future of the market.
For more insights, please visit, https://www.datamintelligence.com/
Found it interesting?
Sai Kiran
Sales Manager at DataM Intelligence
Email: [email protected]
Tel: +1 877 441 4866
We have 5000+ marketing reports and serve across 130+ countries
#interspinousspacersmarketmarket, #interspinousspacersmarketsize, #interspinousspacersmarketoutlook, #interspinousspacersmarketshare, #interspinousspacersmarketforecast, #interspinousspacersmarketanalysis, #interspinousspacersmarkettrends, #interspinousspacersmarketgrowth,
